There is a tussle between those that believe that our COVID vaccines are net beneficial and those that argue that there are too many adverse side effects. The combatants cite data from varying sources, always pointing out that their opponents’ data are, as Mark Twain states, “lies, damn lies, and statistics.” A new paper from JAMA Network Open puts some context to the numbers.
The study is a meta-analysis of 12 studies of all forms of the COVID vaccines. The studies included an inert placebo and provided data of adverse effects in both the study's treatment and control and placebo arms. The studies considered early less severe adverse events, which were separated into categories reflecting the first and then the second vaccination. Here is the picture that tells the story.
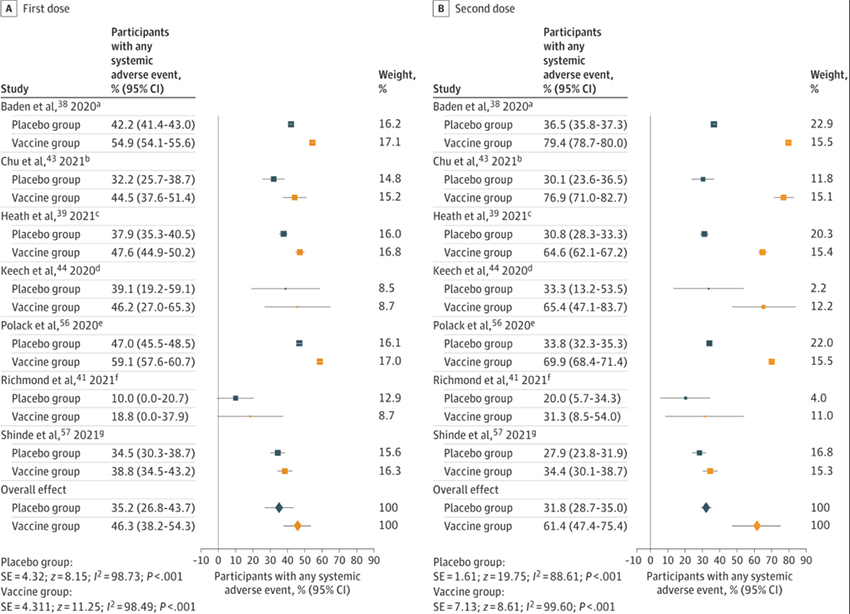
There were more adverse effects in the treatment arm (the orange marks) than in the placebo control groups (the blue marks) in every study but bear in mind that there were also large numbers of adverse effects in the placebo group. By the researchers accounting, adverse effects occurred 75% as often after the first vaccine dose in the placebo control and 52% as often in that group after the second dose —the most common adverse effect, headache or fatigue. Those changes are evident in the distance between the blue and yellow markers in each study.
Here are the take-away thoughts:
- First, look at how strong the placebo effect can be.
- Second, when considering numbers in scientific articles, you must see the comparator to understand the context. The act of participating in a study of an investigational vaccine may heighten your anxiety and stress and can result in a placebo effect. Not discounting that effect when you look at the treatment arm means, at least in this case, that you overestimate the vaccines’ adverse effects.
Source: Frequency of Adverse Events in the Placebo Arms of COVID-19 Vaccine Trials A Systematic Review and Meta-analysis JAMA Network Open DOI: 10.1001/jamanetworkopen.2021.43955