

How about an invention that will benefit everyone?
They don't come along very often, but with a little luck Dr. George Bonheyo (1) and his group at the Department of Energy's Pacific Northwest National Laboratory (PNNL) might have something very big on their hands—a novel way to clean up oil spills, with the aid of chemically modified sawdust.

The esterification of sawdust with vegetable oil. Note the change in volume as the reaction progresses over 8 hours.
Photo courtesy of George Bonheyo.
Researchers have been looking for materials capable of cleaning up oil spills for some time, but with little success. Such a product would have to be non-toxic, absorb oil readily, able to convert it into a floating, collectible mass, be easy to make, and preferably burnable. It would also have to work in icy, rough regions of the ocean. Sawdust itself doesn't do the job but is aided by a very simple chemical reaction called esterification.
Esterification is the process that combines two organic molecules—one a carboxylic acid and the other an alcohol into a single molecule called an ester. Although it is a very simple reaction, esterification can accomplish much. For example, when drinking alcohol is reacted with butyric acid, you get something very different.
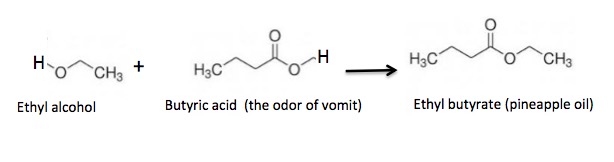
How does this apply to sawdust? To answer that, we need to look at its chemical structure. You might want to be holding onto something first.
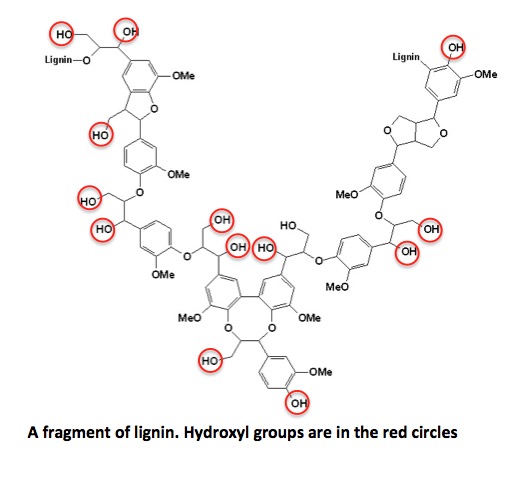
Although lignin looks like a nightmare, chemically it doesn't behave all that differently from plain old alcohol. This is because it has a very large number of hydroxyl groups (red circles). Each of these (theoretically) can be esterified with carboxylic acids, in this case, fatty acids. Shown below is the structure of a very small fragment of lignin, both natural and esterified.
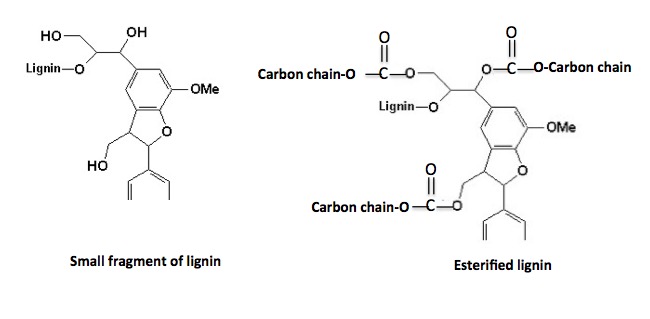
The addition of the fatty acids to lignin significantly changes its properties:
- Like dissolves like. The fatty acid chains act as a sponge to pick up the oil.
- The fatty acids open up the pores of the lignin, allowing more oil to be adsorbed.
- The complex is more biodegradable than is oil itself.
- The complex burns better than oil itself.
- The complex holds together and is easier to collect.
- It is extremely buoyant. It will float for months. Oil can sink.
Bonheyo and his group are discovery scientists, which means that they do not make large batches of the product, but they have a contract with a facility that can make kilograms of material at a time. Previous tests were conducted at the US Coast Guard and Naval Research Laboratory's Joint Marine Test Facility (JMTF) in Mobile, AL and are now ongoing at PNNL’s Marine Science Laboratory in Sequim, WA.
If everything goes well, Bonheyo envisions forming a partnership with a chemical company to make the large quantities that will be needed, and the next time there is an oil spill, perhaps we'll see a crop duster flying over the spill—the method that would be employed to distribute the sawdust.
A very clever idea based on simple organic chemistry. We wish PNNL good luck. You should too.
Notes:
(1) Dr. Borheyo is a Senior Research Scientist at PNNL and a Research Professor of Bioengineering at Washington State University



