My colleague, Dr. Chuck Dinerstein, a retired vascular surgeon and one very sharp guy, now has the misfortune of sharing an office with me. Poor guy. If you pray, pray for him.
But, it has already paid off. We were discussing (what else?) management of postoperative pain, opioid medications to manage it, and a way to do away with the meds altogether (1). It turns out that there is a place for both methods. In certain cases, patients who undergo a painful surgical procedure can be perfectly comfortable without any pain medication, and this can actually get them out of the hospital one day earlier (2,3) because of nerve blocking drugs - local anesthetic drugs injected in the right place.
The non-drug approach is very clever and takes advantage of the sciences of both chemistry and formulation research. It's a little overwhelming to look at but stay with me. It's not as bad as it seems.
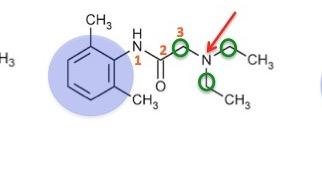
To understand what's going on we need a mini (and presumably dreaded) chemistry lesson on local anesthetics (Figure 1).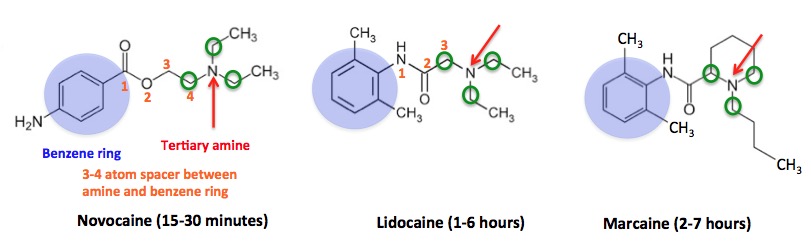
Figure 1. The structure, pharmacophore, and duration of action of three common local anesthetics.
There's a lot going on in Figure 1, but it's not that difficult to understand. The pharmacophore (the portion of the molecule responsible for its pharmacological effect) consists of a benzene ring (blue circle), which is 3-4 atoms (orange numbers, also called a spacer) away from a tertiary amine—a basic nitrogen atom (red arrow) that is bound to three carbon atoms (green circles). Think of it as ammonia in which all three hydrogen atoms have been replaced by methyl groups). While all three share the same pharmacophore, there is a profound difference in the duration of action. Once again, chemistry (Figure 2).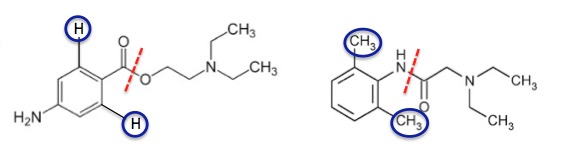
Figure 2. Flanking groups slow metabolism, which leads to a longer duration of action
Figure 2 shows one of many "tricks" that organic chemists use to control drug metabolism. There are a number of ways that these drugs are metabolized. One of them involves an enzymatic cleavage of the bond that is shown by the red hatched line. Once this bond is broken the metabolite will no longer be an anesthetic. Note that Novocaine (left) has one hydrogen atom on either side of the carbonyl (C=O) group—the site of cleavage. But in Lidocaine, these hydrogen atoms are replaced by methyl groups (4), which are also referred to as flanking substituents. The methyl groups fill up a lot more space than the hydrogen atoms and this restricts access of the enzymes (called liver amidases) that break that bond, which prolongs the half-life of the drug (5). This is an example of what we call steric hindrance.
Steric hindrance also explains why Marcaine is a long-acting local anesthetic. The molecule still terminates in a tertiary amine, but one with a very different structure. The two ethyl (2-carbon) groups in Lidocaine are gone; one is now a four-carbon butyl group, and the other is incorporated into a six-carbon ring (a piperidine ring). Both these modifications protect the molecule from metabolism, which is why Marcaine works for so long.
But chemistry can only do so much. The formulation of a drug can have a great impact upon its half-life. And when Marcaine is formulated with liposomes, things get better fast. It's rather amazing. By "packaging" Marcaine with liposomes—microscopic structures which are made up of a phospholipid bilayer that surrounds an aqueous core (Figure 3). Liposomes are great little structures for holding onto things and releasing them slowly. Like drugs.
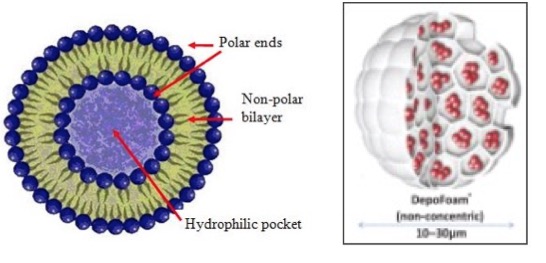
Figure 3. The structure of a liposome (left). A cross-sectional diagram of a DepoFoam liposome containing Marcaine. The Marcaine (red clusters) sit in tiny pockets of water in the liposome. Source: Journal of Pain Research
Pacira, a New Jersey-based company that specializes in putting drugs into liposomes did just that. In 2011 the FDA approved EXPAREL, Pacira's liposomal Marcaine formulation, for the treatment of postoperative pain (6). Marcaine itself is no slouch when it comes to long-lasting pain relief, but the controlled release properties of EXPAREL make quite a difference. EXPAREL was effective for up to three days, compared to 7 hours for Marcaine. How well does it work? According to Dr. Dinenstein, very well. In 2012 Dinerstein began using the product off-label by injecting it directly into the wound while closing. About 95% of his patients had their pain controlled well enough that they didn't need any pain medications.
Combining a drug that was discovered in 1957 with new formulation technology provides an alternative to post-operative pain and the drugs needed to treat it. Pretty impressive, no? (6)
You can read Dr. Dinerstein's companion piece about the use of long-term anesthetics here.
NOTES:
(1) Post-surgical pain management can be complicated and the meds can cause problems, which may require a longer stay in the hospital. Self-administered morphine is generally considered the best method, not pills.
(2) Don't worry. I'm not calling for opioid restrictions in hospitals (or anywhere else). But if you really don't need them why take them?
(3) Dr. Dinerstein used this non-opioid approach during hundreds of surgical procedures over a two-year period. It worked very well.
(4) A direct comparison of Novocaine to Lidocaine is unfair because the former contains an ester linkage from the benzene ring to the rest of the molecule. Esters are metabolized much more easily than amides. This example is just meant to demonstrate how molecular structure and metabolism are closely linked.
(5) The metabolic fate of these drugs is far more complicated than what I showed. In fact, the first metabolic step is an enzymatic removal of one of the ethyl groups of the tertiary amine followed by the breaking of the amide bond.
(6) It is science, not bureaucratic idiocy, which will provide better alternatives than opioids for pain management. In the meantime, they are the best choice. Let the bureaucratic idiots and sycophants who are responsible for current patient misery go away somewhere. Far.



