At the American Council, we specialize in taking complex scientific and medical issues and crafting them into a form that can be understood by non-scientists, while at the same time not omitting crucial information. And talk about timing! We don't get a challenge like this every day. This one is special. Do people pee in the pool? How about bears? We shall see.
The world can be broken down into two broad categories:
1) Those who deny that they pee in the pool.
2) Those who won't go in the pool because they know that the people in group #1 are a bunch of liars.
Given these two facts, it is difficult to understand why anyone would want to study whether urine is found in pools, let alone how much. But, a paper in Environmental Science and Technology Letters redefines the term "yellow journalism."
The study consisted of determining the amount of urine in a typical pool by measuring tiny amounts of the little-used artificial sweetener acesulfame potassium—a surrogate marker (1). Results: 100 percent of pools are peed in. They try to dress it up with science:
"Nitrogenous organics in urine can react with chlorine in swimming pools to form volatile and irritating N-Cl-amines. A urinary marker is desirable for the control of pool water quality."
OK, this is nuts. N-chloroamines are formed (in miniscule quantities) when urea reacts with chlorine; which is also volatile and irritating. Which is worse? Neither. Not in the concentrations that were detected (1,2). People have been swimming in (and peeing in) pools forever. Yet, we all live another day, presumably to pee in another pool.
"The widespread consumption of acesulfame-K (ACE), a stable synthetic sweetener, and its complete excretion in urine makes it an ideal urinary marker (3). Here we report the occurrence of ACE and its potential application in swimming pools and hot tubs [aka herpes spawners]."
"[U]p to 570-fold greater than in the input tap water."
Well, that should certainly stop all of you who would otherwise intentionally gulp down pool water. And if you're heavily invested in the bottled pool water industry, your life just went down the urinal.
If these guys can get paid to study something that every multicellular organism in the universe already knows, then I feel justified in proposing a couple of other studies. They are just as valid.
1. What comes out of a toaster?
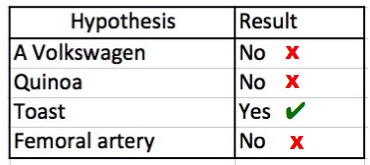
Protocol: 1) Collect toaster gas; 2) Inject into a quadrupole mass spectrometer; 3) Download a database of all known matter; 4) Compare toaster data using a Cray XT4 supercomputer cluster; 5) Record results.
Images: Wikimedia Commons, Wikimedia Commons
And, here are the results:
2. Which is hotter? The Kilauea Volcano or mayonnaise?
Protocol: Dip Dr. Oz's head into each. Record results.

Images: Wikimedia Commons, Flickr
Results: The Kilauea Volcano is hotter than mayonnaise:

Image: Serious eats
Addendum: Kilauea Oz's next show was unremarkable. The content was similar to most of the others, and the audience did not notice a difference. Mayonnaise Head Oz's episode has not aired yet. I suggest that you hold the mayo.
OK, I've wasted five minutes of your time. Guilty as charged. But at least I didn't collect 250 waste samples from 31 pools and tubs in Canada to look for pee that was guaranteed to be there.
At the very least, I can answer my own question...

Notes:
(1) ACE makes up about 10 percent of the artificial sweetener market in the US. It was chosen as a surrogate marker for pool urine because it is chemically stable. Aspartame would be a more logical choice because of its wide use, but it doesn't even survive your stomach let alone your bloodstream blood. Because of metabolism, there is none in the urine.
(2) The concentration of ACE measured ranged from 30 to 7110 ng/L. To put this in perspective, 30 nanograms is 0.00003 mg. A grain of salt weighs 0.3 mg. So, at the lowest measured concentration, it would take 10,000 liters, or 2,640 gallons of pool water (a quarter of the pool) to get the same amount of ACE that would equal the weight of one grain of salt in one liter of water.
(3) The real science in this paper is the power of analytic chemistry, specifically the ability of modern instrumentation to measure supernaturally low concentrations of chemicals. Ironically, this advance in science has also created an increase in anti-science. Most phony chemical scares arise from the detection of chemicals that could not be detected before but were there all along. And doing no harm.




