
"Organic" may be one of the most confusing words in language today. Although most people will probably have a vague idea of the meaning of the term they probably won't know the definition. Part of the confusion arises from the fact that there are two, very different, definitions for the same word, one from chemistry and one from agriculture. The chemistry definition is unambiguous and clear-cut, at least most of the time. Let's start here.
THE CHEMICAL DEFINITION OF ORGANIC
In chemistry, the definition is based solely on chemical structure. With very few exceptions a chemical is classified as organic if it contains at least one carbon atom, regardless of its source. This is why organic chemistry is called "the chemistry of carbon."
But there are exceptions. Carbon dioxide certainly contains an atom of carbon but is classified as inorganic. Common inorganic chemicals include salt, ammonia, baking soda (sodium bicarbonate), and sulfuric acid. But, like carbon dioxide, baking soda also contains a carbon atom yet is still classified as inorganic. What's going on? For a chemical to be organic there is an additional requirement. A hydrogen atom must be chemically bound to a carbon atom. Figure 1 demonstrates examples of carbon-containing chemicals, some of which are organic and some that are not.
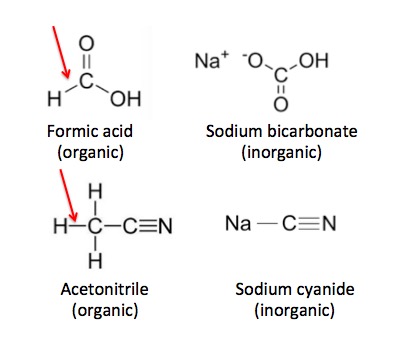
Figure 1. Organic vs. inorganic. Formic acid (top) is organic because it contains a carbon-hydrogen bond (red arrow) but sodium bicarbonate, although similar in structure is inorganic because it lacks this bond. Acetonitrile and sodium cyanide (bottom) is another example of the same rule.
But in organic chemistry, nothing is ever entirely straightforward. Both carbon tetrachloride and urea (1) (Figure 2) are considered to be organic, despite the fact that neither molecule contains a carbon-hydrogen bond. These exceptions arise from historical precedent.
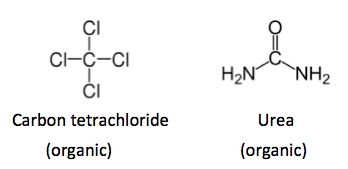
Figure 2. Both carbon tetrachloride and urea are usually considered to be organic. Neither chemical has a carbon-hydrogen bond. This is a rare exception.
THE AGRICULTURAL DEFINITION OF ORGANIC
The agricultural use of the term is entirely different from the chemical use. It is a definition of which practices are permitted for food so that it can receive the UDSA Certified Label. According to the USDA:
"Organic meat, poultry, eggs, and dairy products come from animals that are given no antibiotics or growth hormones. Organic food is produced without using most conventional pesticides; fertilizers made with synthetic ingredients or sewage sludge; bioengineering; or ionizing radiation."
The list of pesticides that are permitted in organic farming can be found here. This brings up an interesting paradox, as shown in Figure 3.
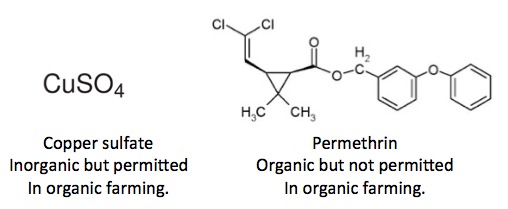
Figure 3: Clash of the terms. Copper sulfate is an inorganic chemical but is approved for use in organic agriculture. Permethrin is an organic chemical, which is used in conventional agriculture but is not permitted in organic farming.
Chemistry can sometimes be profoundly confusing. This is one of those times.
NOTE
(1) Urea, which was discovered in 1773 by a French chemist, was the first "organic" chemical ever isolated. It is considered to be organic even though the molecule does not contain a carbon-hydrogen bond.



