This flu season is turning out to be very bad, something that my colleague Dr. Julianna LeMieux has been covering. (See: here and here). Unfortunately, influenza is difficult to avoid, prevent or treat. This year's vaccine is (at best) moderately effective, and the alternative - flu drugs - is not great either. Even so, there are now shortages of these medicines, despite their shortcomings. In reality, there are simply no wonderful choices for either preventing or treating flu.
Nonetheless, in response to this especially nasty circulating strain, called H3N2, the CDC has recommended the use of antiviral influenza drugs, mainly Tamiflu (1) (generic name oseltamivir). Oseltamivir, which was approved by the FDA in 1999, may not be a great drug, but given the severity of this year's flu, and the fact that oseltamivir use has skyrocketed, it's not a bad time to examine its pluses and minuses, something I have written about before (2). Following is the summary of a systematic Cochrane review from 2014 about the safety and efficacy of oseltamivir. (NOTE: It is critical to take flu antivirals as early as possible in the infection. After about two days they won't help. This is analogous to starting Valtrex immediately after the first signs of a herpes outbreak).
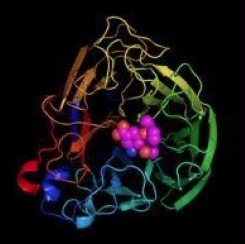
An x-ray crystallography depiction of oseltamivir bound to the active site of neuraminidase. Image: Graphics Buzz
In a 2014 review entitled "Neuraminidase inhibitors (NIs) for preventing and treating influenza in adults and children," Tom Jefferson an epidemiologist at the University of Oxford and colleagues from five other centers examined studies of oseltamivir and its "chemical cousin" Relenza (generic name zanamivir) to "describe the potential benefits and harms of [neuraminidase inhibitors] for influenza in all age groups by reviewing all clinical study reports of published and unpublished randomised, placebo-controlled trials and regulatory comments." The group chose not to include journal papers because "in previous versions of this review we identified unresolved discrepancies in the data presented in published trial reports and substantial publication bias."
Rather than using journals, the group instead gathered data from regulatory agencies in the UK, US, Japan, and the European Medicines Agency as well as two manufacturers of the drugs, GlaxoSmithKline and Roche. This enabled them "to verify information from the randomised, placebo-controlled trials on adults and children with confirmed or suspected exposure to naturally occurring influenza."
Here are the findings (I have focused only on Tamiflu/oseltamivir because it is most widely used):
- Adults who took oseltamivir began to feel better in 6.3 days while those who did not feel better in 7 days.
-
The same trend was seen in children, except for those with asthma, where no effect was seen.
-
Oseltamivir did not significantly reduce hospitalizations for either adults or children.
-
Oseltamivir reduced the spread of influenza in households.
-
Oseltamivir reduced self-reported (but unverified) pneumonia, but this effect was not seen in five trials that used better diagnostic criteria for pneumonia. There were no studies that examined any effect on radiologically-confirmed pneumonia.
-
Nor did oseltamivir show any benefit in preventing other serious influenza complications.
-
Oseltamivir did not reduce otitis media (ear infection), sinusitis or bronchitis in adults or either adults or children.
-
In adults, oseltamivir increased the risk of nausea and vomiting but decreased the risk of diarrhea. In children, oseltamivir increased vomiting.
-
There was an elevated risk of neuropsychiatric adverse events (NPAEs) although a review in Drug Safety contradicts this: "The available data do not suggest that the incidence of NPAEs in influenza patients receiving oseltamivir is higher than in those who do not, and no mechanism by which oseltamivir or oseltamivir carboxylate could cause or worsen such events could be identified."
Based on the Cochrane findings it is clear that oseltamivir is a highly imperfect drug.
"The balance between benefits and harms should be considered when making decisions about use of both NIs for either the prophylaxis or treatment of influenza. The influenza virus-specific mechanism of action proposed by the producers does not fit the clinical evidence." (5)
Of course, there have been other reviews of the efficacy and safety of NIs. Two more recent reviews present a more sanguine picture.
A 2015 Lancet review by Dobson, Whitley, et. al. examined nine clinical trials including 4,328 patients. The authors noted:
- There was a 21% shorter time to alleviation of all symptoms (oseltamivir) compared to placebo (97.5 hours vs. 122.7).
- Oseltamivir reduced respiratory tract complications requiring antibiotics (4·9% oseltamivir vs 8·7% placebo).
- Oseltamivir reduced admittances to a hospital for any cause (0·6% oseltamivir, 1·7% placebo).
- As noted in the Cochrane review the drug caused nausea and vomiting, but no significant neurological or psychiatric effects were noted.
"With influenza being as bad as it is this year, discussion of neuraminidase inhibitors is critical. The bottom line is therapy needs to be initiated early in the course of illness and must be given to high-risk individuals."
Richard Whitley, M.D., Division Director Pediatric Infectious Diseases, University of Alabama Birmingham.
Also, a 2017 review by Malosh, et. al., found a similar effect (and side effects) in children:
"Despite substantial heterogeneity in pediatric trials, we found that treatment with oseltamivir significantly reduced the duration of illness in those with influenza and lowered the risk of developing otitis media. Alternative endpoints may be required to evaluate the efficacy of oseltamivir in pediatric patients with asthma."
Malosh, Whitley, et. al., Clinical Infectious Diseases, November 2017
Although there are differing opinions about the utility of anti-flu drugs, they are imperfect by any measure. It's much better to prevent flu than to treat it. Better roll up your sleeve.
NOTES:
(1) Three NI flu drugs are approved. Tamiflu (generic name oseltamivir) is widely used. The other two are Relenza (zanamivir) and Rapivab (peramivir). All three work by the same mechanism.
(2) See: Tami-flu the Coop? (2012)
(3) Neuraminidase is one of two enzymes that are involved in viral infectivity. Hemagglutinin enables the virus to enter the host cell. Neuraminidase enables newly formed virus particles to break free from the host cell.
(4) Tamiflu is a pill. Relenza is an inhaled powder. Rapivab is an IV solution.
(5) This suggests that Tamiflu and Relenza may be operating by a mechanism that is different from the antiviral inhibitory mechanism that is expected. I don't buy it. Not one bit.