
Causes of obesity are not as simple as a lack of exercise or overindulging. It has been known for some time that a predisposition for obesity has been linked to certain genes, however, specific mechanisms have been more difficult to elucidate.
Some changes in our DNA, called mutations, alter the sequence of the bases in our DNA which can result in changing a trait or a disease. Other changes in our DNA - epigenetic changes (changes to our DNA that do not have to do with the sequence of bases) - can lead to the same result without the DNA sequence being changed. In this process, DNA gets marked with epigenetic 'tags' that can make the DNA alter its conformation. The result is that genes are expressed more or less which has an effect on the amount of proteins that they produce.
Recently, a study published in EBioMedicine compared the amount of epigenetic tags (more specifically, methyl groups) in the DNA of babies at the time of their birth (using DNA from the umbilical cord) with the amount of fat tissue in the same children at the age of six years old.
They focused in on a part of one gene called CDKN2A - a gene involved in making fat cells. They found that lower DNA methylation at the CDKN2A gene was associated with a greater risk of the child developing obesity in later life.
That is important because low amounts of methylation, in general, correspond to a more active gene. So, low amounts of methylation indicate that the gene CDKN2A is more active. When that gene is responsible for making fat cells, it is clear to see how that person could be predisposed to obesity.
The data below show some of the results. In this graph, the colored points indicate the person's percent body fat (red = lowest percent body fat; blue = medium percent body fat; green = highest percent body fat.)
What can easily be seen is that the group with the highest percent body fat (green) also had the lowest percent methylation and that the low and middle percent body fat had consistently higher methylation.
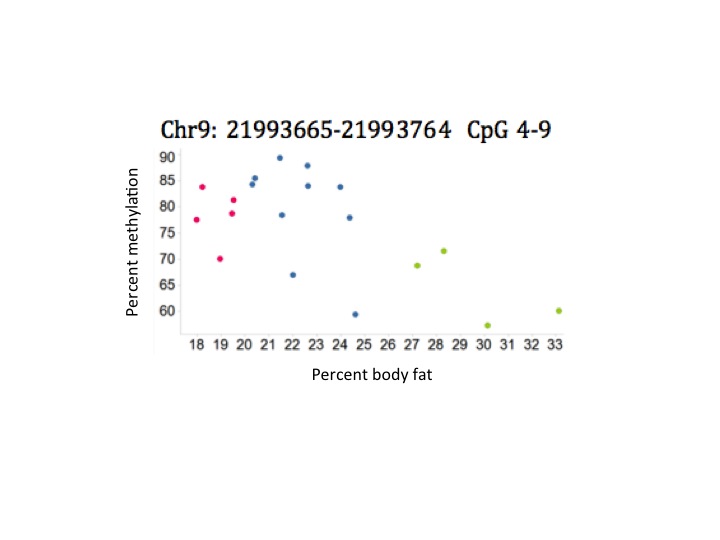
In fact, a 10 percent decrease in methylation at the CDKN2A gene was associated with an increase in fat mass of around 220 g (0.5 pounds), at age 4.
More than 1 in 3 adults and 1 in 6 children are categorized as obese in the United States. Awareness of early risk factors could identify people early so that early interventions can be put in place to combat their genetic predisposition.
The study went beyond just looking at DNA at birth. They also compared the methylation of DNA in this region in fat tissue of obese and lean adults and found that methylation of CDKN2A was lower in the fat tissue of obese adults when compared to lean adults.
This work, which provides evidence of an epigenetic profile present at birth being linked to obesity, is important on many levels. It may lead to early identification of people predisposed to obesity. Because epigenetic marks are able to be altered through the environment, pregnant mothers could modify their habits to change the methylation of their babies' DNA, although just how they could do is, as yet, unknown. Lastly, this provides information about the role of the CDKN2A gene in adult obesity and could lead to future adult weight loss therapies.
References:
* Lillycrop, K., et al. A Perinatal Marker for Later Adiposity. Ebiom (2017), doi: 10.1016/ j.ebiom.2017.03.037
* https://www.niddk.nih.gov/health-information/health-statistics/overweigh...



