There are many questionable aging-related products on the market. One of the more recent is TeloYears, a test that supposedly determines your "true age" by measuring the length of a structure on your chromosomes called telomeres.
Telomeres can be thought of as a protective cap. Each time your cells divide, the telomere caps on the DNA that makes up your chromosomes shorten just a bit. When the telomeres get too short, the cell dies. If your telomeres are shorter than they ought to be, the thinking goes, then your body is actually "older" than your chronological age would suggest. On the flip side, if your telomeres are longer than they ought to be, then your body is "younger" than your chronological age would suggest.
Is this test accurate? Probably not. There doesn't seem to be any credible link between telomere length and overall health. According to Genetic Literacy Project, there may not be any clinical value to measuring telomere length whatsoever.
But that doesn't mean that all investigations into aging-related biomarkers are doomed. New research published in the journal Nature Biomedical Engineering shows that molecular and physical changes in skin cells can be used to calculate a "cellular age" that may be used as a proxy for healthspan.
The Skin Doesn't Lie
The authors examined dermal fibroblasts (skin cells that produce connective tissue) in 32 samples taken from people who ranged in age from 2 to 96 years old. The cells were subjected to nine tests that measured biophysical characteristics (such as a cell's ability to move and to exert forces on its surroundings) and biomolecular characteristics (such as the size of a cell's stockpile of ATP -- the "energy currency" -- or its ability to repair damaged DNA).
As predicted, the scientists were able to tell the difference between fibroblasts taken from young people and old people. For instance, cells from older people were larger, stiffer, less mobile, and exerted greater traction forces than cells from younger people. Furthermore, older cells contained less ATP and were not as adept at repairing DNA damage. Additionally, cells from older people were more heterogeneous than cells from younger people; in other words, older tissue samples had cells with highly variable characteristics, while younger tissue samples had cells with more uniform characteristics.
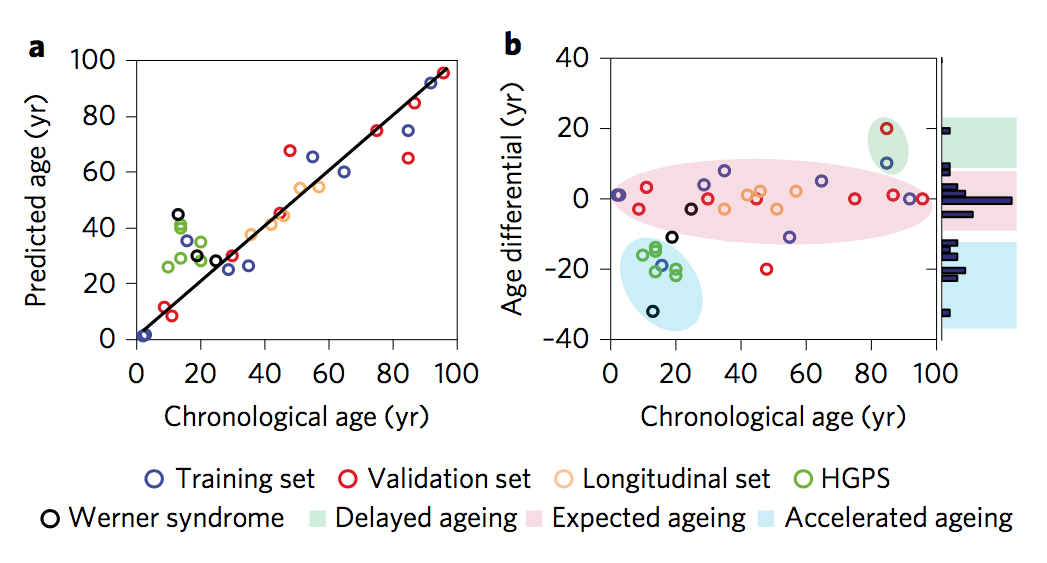
Armed with this mountain of data, the authors then examined if they could use it to predict the age of a donor simply by examining his or her skin cell characteristics. Indeed, they could. Using merely two parameters (cell roughness and the size of cellular nuclei), the authors could determine the age of the donor, albeit with a slight error of about 6 years.
Finally, the authors could use their model to detect outliers and abnormalities. By plotting the age differential (chronological age minus predicted biological age) versus chronological age, they could determine if a person's cells were older or younger than they should be. Importantly, their model was also fairly accurate at detecting tissue samples taken from people with genetic diseases (Hutchinson-Gilford progeria syndrome and Werner syndrome) that cause premature aging.
If the authors' results hold up under further scrutiny, the implications are potentially large. Aging is not well understood, but this team's results may provide several important leads into the molecular mechanisms of aging. This could allow for the development of drugs that target chronic diseases, as well as therapies to help mitigate the inevitable physiological decline associated with growing old.
Source: Jude M. Phillip, et al. "Biophysical and biomolecular determination of cellular age in humans." Nat Biomed Eng 1, Article number: 0093. Published: 11-July-2017. doi: 10.1038/s41551-017-0093




