Precision medicine, the promise of highly targeted, customized care is so alluring, but much of its hope and attraction is lost in the translation from science to practical care. Two recent studies demonstrate how the hoped-for translation is tantalizingly close and elusive. High blood pressure, hypertension, is the dominant noncommunicable disease globally; and a well-known risk factor for both stroke and coronary artery disease. It has been extensively studied and family studies have shown it to result from both genetic and lifestyle factors.
Genetics
Our friends, the UK Biobank have produced another Genome-Wide Association Study (GWAS) in this instance on hypertension. You can catch up on the Biobank and GWAS studies in general here and here. For those looking for the gist, a GWAS study identifies genes associated with particular outcomes, like hypertension, the need to wear glasses or even doing well in school. Prior studies of high blood pressure have suggested/identified 274 genetic alterations accounting for 3-4% of patients with hypertension.
The UK Biobank coupled additional data from the US Million Veteran Program and Estonian Genome Centre Biobank expanding the study population to over 1 million individuals, resulting in a 3-fold increase in identified loci and increasing the genetic “explanation” of hypertension to 11%. As with any genetic study, the findings are restricted to the gene pool, in this instance, Europeans or their descendants. Hypertension was defined by elevated systolic or diastolic blood pressures as well as pulse pressure or pulse width. [1]
As with most GWAS studies, the researchers developed a composite genetic risk score. Taking all 901 loci into account generated scores that demonstrated a 10mmHg elevation between the highest and lowest population segments, in this instance quintiles. They extrapolated that 10mmHg rise in blood pressure between the lowest and highest quintiles into clinical events demonstrating that their genetic risk score identified patients at increased risk of stroke (37% higher) and high attacks (44% increase). [2]

Some genes were associated with one component of hypertension, others with multiple – as the overlap in this Venn type diagram demonstrates. The identified genes had roles in modulating vascular tone, the constriction, and dilation of blood vessels as well as genes involved in adrenal tissue – which has a role in fluid retention through the production of aldosterone. These pathways were already known or suspected. But there were other surprising genes associated with hypertension including genes involved in the remodeling of vessels, reshaping the heart wall, sodium handling by the kidneys, as genes associated with type 2 diabetes, elevated cholesterol, and atherosclerosis. The chart below shows the multiplicity of interactions of these "hypertension" genes with other diseases. Epistasis is a term used to describe the impact of alterations of one gene upon another. The combinatorial possibilities of these interactions are enormous.
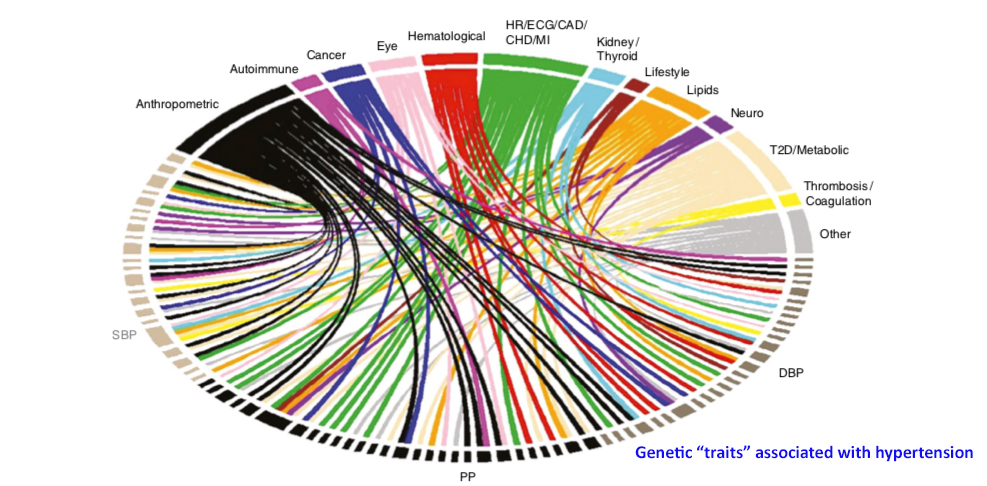
The variability in the expression of the phenotype, a genetic way of saying a patient has hypertension, by genes accounts for only 11% of cases in this study - a common percentage in the GWAS literature. When the same risk score was applied to an Asian population, its predictive value dropped, to about 6%; not surprising given that Asian and European genetic variations would be expected to be similar, not identical. This study shows that many hundreds of genes are involved and interact in the expression of the phenotype we call high blood pressure. Not only is there no easily defined “genetic basis” for hypertension, but the combinatorial possibilities are so great as to be considered, at this point, infinite. Given our current understanding these genetic variants are better described as a “background effect,” a necessary but not sufficient “cause” of hypertension.
Environment
Scientists have investigated the “background effect” of genes on phenotypes and have found, not surprisingly that in addition to multiple genetic variants interacting with one another, they also respond to their environment. Understanding the added complexity of environmental or in the case of medicine lifestyle factors along with genetic variation is a very tough knot to untie and untangle. Research on a much simple biological model, yeast helps outline the bigger picture.
Researchers investigated genetic background effects in yeast, looking at seven genetic variants in 10 various environments. [3] They identified over 1000 epistatic genetic interactions for those seven genetic variants. Some alterations affected 100 or so other genes, other impacted over 500. And the interactions resulted in both enhanced and reduced phenotypic expression. In their words, phenotypic expression was “complicated, highly contextual.” And the context was the yeast’s environment, which accounted for 50% of expression in this study. Their findings echo the UK Biobank’s hypertension report on a far smaller scale. A back of the envelope calculation places the possible interaction of hypertension's 900 loci at well over 100,000 possibilities; and the number of our environments, based on diet and activity alone is far greater than 10.
Precision medicine is an aspirational, not pragmatic goal. These two studies, taken together make it clear that the possible genetic and environmental interactions underlying the phenotypes we call disease are not going to result in one or even a few medical answers. How and where we live our lives are the only variables we can control. The deeper we look into our biology, the more precise our findings, the more we realize that health results from our choices in the world, not from some preordained genes.
[1] For a BP of 120/80 the pulse width is 40. Systolic pressures are frequently due to fluid shifts while diastolic pressures frequently correlate with the constriction or dilation of blood vessels. Pulse pressure describes the difference between systolic and diastolic pressures and is a more subtle measure of what is driving the underlying pathophysiology.
[2] The term extrapolation is critical, these increases were predicted based upon mathematical formulas relating blood pressure increases to morbidity. No actual observations are represented by these statements.
[3] Environments included the addition of various antibiotics, ethanol, temperature changes, peroxide and glucose. “different carbon sources or temperatures or chemical challenges.”
Sources:
Genetic Analysis of over 1 million people identified 535 novel loci for blood pressure Nature Genetics DOI: 10.1038/s41588-018-0205-x
The Complex Underpinnings of Genetic Background Effects Nature Communications DOI: 10.1038/s41467-018-06023-5



