There is no shortage of blame for the microbial mess we now find ourselves in. Antibiotics that used to easily cure infections no longer work, leaving us dangerously close to the pre-penicillin days. In the absence of new antibiotics (especially new classes), this trend can only continue. But, thanks to an observational study conducted by multiple groups in the UK, it may be possible to buy us some more time by using antibiotics more judiciously. They even tell us how.
Some of the "culprits" that have put us in this situation include:
- Evolution (selective pressure): It is inevitable that resistant bacteria or viruses will emerge when they are exposed to a drug that kills them, or inhibits their growth.
- Drug companies: Companies have been blamed for abandoning antibiotic research for more lucrative therapeutic areas, such as oncology.
- The FDA: In the 1990s, ill-advised (and pointless) changes that were pushed by statisticians were supposed to increase the statistical power of clinical trials of new antibiotics. This made it prohibitively expensive, time consuming, and sometimes impossible (1) for companies to conduct trials. It is no coincidence that all major pharmaceutical companies abandoned antibiotic research at this time.
- Inadequate hygiene and sanitation in hospitals
- The use of antibiotics as growth promoters in livestock feed
- The misuse of antibiotics in people, especially prescribing them when they are not needed, e.g., for viral infections.
Although there have been reports of infections that are resistant to all antibiotics, arguably, the biggest threat to public health at this time is drug resistant Clostridium difficile (C.diff.)
Infection by Clostridium difficile (2) can arise when antibiotics, especially broad spectrum antibiotics, wipe out the bacteria that are normally present in our gastrointestinal tract. The absence of normal bacterial flora allows the C. diff. to overgrow and take over. Clostridium is especially dangerous because it produces toxins that damage the lining of the intestines, resulting in diarrhea, which can be severe, and inflammation, which can be lethal. About 500,000 people per year in the US become ill from C. diff, and 30,000 will die from it. Worse still, more aggressive strains of the bacterium that produce larger amounts of toxins have become more common. The graph below illustrate the growing threat of C. diff. to human health.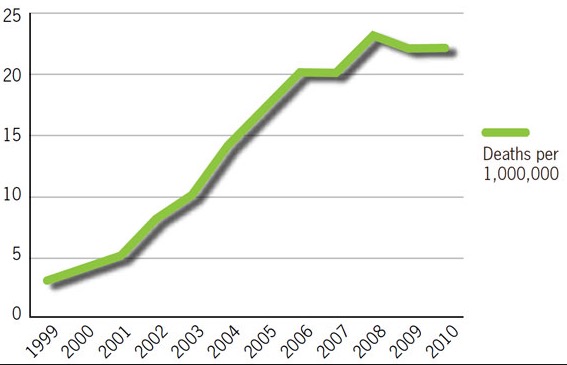
Deaths in the US caused by C. diff. 1999-2010 Source: CDC
C. diff. is also difficult to get rid of, because most antibiotics don't kill it (3,4). The emergence of more aggressive strains of the bacterium prompted the CDC to name it "the most urgent of all superbug infections" in 2013.
While all of these factors have played a part in the emergence of resistant bacteria, the relative importance of each factor is not clear. The British study, which was just published in The Lancet shed some light on the cause of a 2006 outbreak of the dangerous superbug— drug resistant C. diff. in the UK. The culprit in this outbreak was the use (or overuse) of a class of antibiotics called fluoroquinolones (2). This can be seen in the graph below.
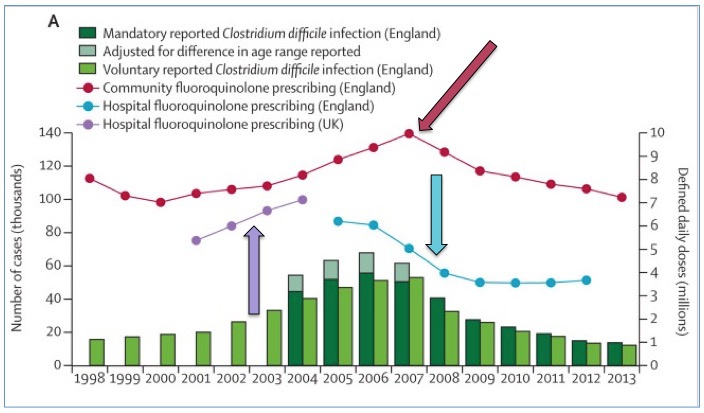
The timeline of the 2006 C. diff. outbreak in the UK. Source: The Lancet
The dark red line represents the number of non-hospital prescriptions between 1998-2013. The red arrow points to 2007—the year during which the highest number of fluoroquinolone prescriptions were written. The number of infections (green bars) correlates very well with the number of non-hospital prescriptions and also the number of hospital prescriptions in the UK (purple). The turquoise line represents the use of fluoroquinolone in English hospitals between 2005 and 2012.
Note that in the seven years following 2006—the year that fluoroquinolone national control policies were instituted in Britain— that a reduction of hospital prescriptions of fluoroquinolones resulted in an 80 percent drop in C. diff. cases. But, this drop could also be explained by (or entirely due) to improved hygiene in hospitals.The authors considered two possible scenarios that would account for the decrease:
- If improved hospital hygiene was responsible for the reduction of C. diff. cases, then a reduction of all strains of C. diff. across the board would decline.
- If the reduction in fluoroquinolone use was responsible for the C. diff. decrease, then the reduction od of fluoroquinolone-resistant C. diff. strains would decline disproportionately.
The answer is the latter. In the graph below it is clear that limiting fluoroquinolone antibiotics resulted in a large decline in fluoroquinolone-resistant bacteria in isolates (5).
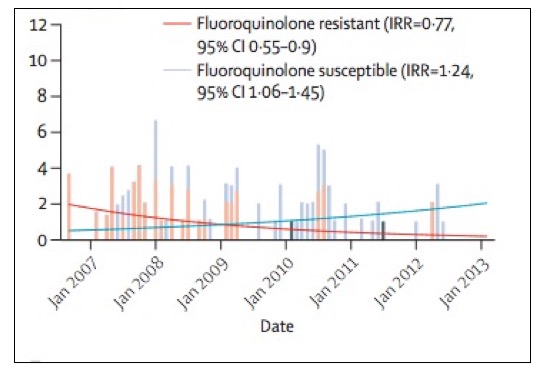
The composition of C. diff. isolates following fluoroquinolone restriction 2007-2013. Source: The Lancet
The good news is that more judicious use of antibiotics will buy us a little more time. The bad news is that we are still running out of time.
Notes:
(1) ACSH advisor Dr. Shlaes noted in 2013 about the FDA's more stringent requirements for antibiotic clinical trials in the 1990s: "The number of patients required has skyrocketed into the thousands, further increasing the cost and the time it will take to run such a trial."
(2) Difficile, is Latin for difficult. The bug is hard to get rid of because it has the ability to survive unfavorable conditions.
(3) Vancomycin, the "go-to" drug for MRSA and other multi drug resistant infections is used orally to treat C. diff. infection. The drug is poorly absorbed and stays in the gut, so it can fight the infection without the systemic toxicity of IV vancomycin. This is the only approved use for oral vancomycin.
(4) Under conditions of stress, C diff. also forms spores. These are even harder to kill.
(5) The increase in fluoroquinolone susceptible cases of C. diff. can be explained by resistance to other classes antibiotics.



