If you read enough phony chemical scares by EWG, NRDC, Joe Mercola and other know-nothings, you may wonder how it is possible that there is a single person left on earth. This is because we are eating so many "deadly" chemicals. But so were our ancestors. These chemicals arise from the ghastly, toxin-forming process called cooking. Yet we're still here. How odd.

Ancient Roman Oven. They ate deadly chemicals too. Photo: Wikipedia Commons
The best known chemical reaction that occurs during cooking is the Maillard Reaction. The Maillard, which is now more than 100 years old, is responsible for foods turning brown during cooking. It is known to produce hundreds, if not thousands, of chemicals, most of which are flavors and odors. The chemical reactions arise from the heat-promoted reaction of amino acids and sugars, both of which are ubiquitous in foods and beverages.

Louis-Camille Maillard. Nice stache. Photo: Wikipedia Commons
Of the myriad of chemicals formed, a few have raised "concern" in the chemical scares industry, especially acrylamide, which elicits the expected response from the usual nincompoops. (1).
"Acrylamide is a chemical linked to cancer that can form in some foods during high-temperature cooking processes."
AgMag (EWG), March 2015
"But did you know [French fries, potato chips and white bread] can also contain a toxic by-product of the cooking process that’s been linked to cancer?"
Dr. Oz, "Toxic Toast? The 411 on Acrylamide"
"Acrylamide is a cancer-causing and potentially neurotoxic chemical and is created when primarily carbohydrate foods are cooked at high temperatures, whether baked, fried, roasted, grilled or toasted."
Crazy Joe Mercola, Heat-Induced Acrylamide May Be a Primary Hazard of Processed Food
In addition to acrylamide, two other chemicals that are formed during cooking have attracted attention. Hydroxymethylfurfural is a suspected carcinogen and 2,3-butanedione (aka diacetyl, biacetyl) is responsible for "popcorn lung.
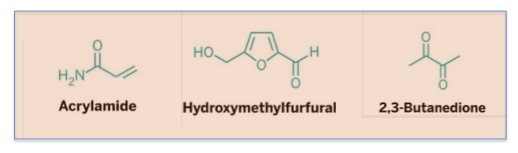
Three "chemicals of concern" formed by the Maillard Reaction Adapted from "The Maillard Reaction Turns 100." Chemical and Engineering News, October 2012.
But it was not Maillard that brought an end to the Roman Empire or anything else. The "chemicals of concern are not all that concerning.
- Acrylamide: We have debunked its risk many times (1).
- Hydroxymethylfurfural: "No relevance for humans concerning carcinogenic and genotoxic effects can be derived. The remaining toxic potential is rather low. Various animal experiments reveal that no adverse effect levels are in the range of 80-100 mg/kg body weight and day. Safety margins are generally sufficient."
- Biacetyl: Yes, it can damage lungs if a sufficient amount is inhaled. But it occurs in a wide variety of foods, including butter, cheese, bread, coffee, brandy, and rum.
The bottom line is that Rome wasn't poisoned in a day (except by lead), and a loaf of bread should be pretty far down on your list of things to worry about. These chemicals have been consumed for thousands of years, yet we still exist.
If you think you're going to get through this thing without... The Dreaded Chemistry Lesson (DCL), think again. The DCL explains why the Maillard Reaction occurs at all. It's simple organic chemistry.
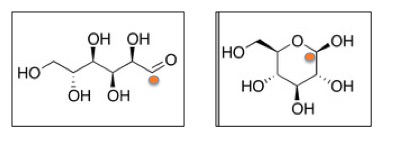
Dreaded Chemistry Lesson, Part 1 - Sugars exist in two forms, which are in equilibrium, so both species are always present. The open form (left) contains an aldehyde (orange circle), which is chemically reactive. The orange circle on the closed form (right) depicts the carbon which is the aldehyde group when the sugar is in the open form.
Dreaded Chemistry Lesson, Part 2 - The aldehyde in the open form (as do all aldehydes) reacts with amines, including amino acids. Since there are 20 amino acids in most proteins and several sugars in carbohydrates, a large number of Schiff Bases can be formed.
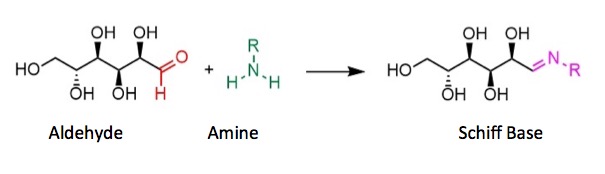
Dreaded Chemistry Lesson, Part 3 - Schiff Bases (purple, above) are chemically unstable. They react with all sorts of things, especially when heated, and even with themselves - a reaction called the Amadori Rearrangement. The many reactions of the Schiff Bases plus the number of different sugars and amino acids present in foods are the primary reason why so many different chemicals are formed during baking. Note that there is no image of the Amadori Rearrangement. This is intentional. It would give you nightmares.
As a special reward for getting through the DCL, I'm going to suggest you treat yourself to a tasty snack, courtesy of Louis-Camille Maillard.
Bon appétit.

Note:
(1) See Dr. Ruth Kava's "European Union Still Drinking The Acrylamide Kool-Aid"




