These are very confusing times for people, especially those without any chemistry training, who are trying to figure out the often confusing and conflicting information about what information toxicology can provide about what drug is responsible for an overdose of "fentanyl." Was the OD caused by fentanyl or one of its analog? This information is important because it can give us information about its origin. Was the OD caused by legal pharmaceutical fentanyl (rarely) or by fentanyl or an analog related to it which was made in a lab in China?
There is a whole lot of confusion and misinformation out there, so I thought it might be useful to give a primer about how analytical chemistry, knowledge of metabolism, and determination of the chemical structure of the of impurities arising from the inadequate purification an illicit drug can tell us quite a bit about its source.
Unfortunately, this cannot be done without some chemistry. My apologies in advance. First, let's look at the breakdown pathways and products of fentanyl (Figure 1). 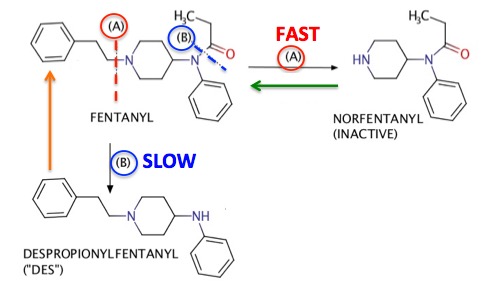
Figure 1. Although fentanyl undergoes a number of metabolic degradations, I have included only the two that best exemplify how chemists can distinguish between pharmaceutical fentanyl from what might be bought on the street. The primary (fastest) metabolism is called oxidative dealkylation, (A) in which the bond that breaks is shown in a red hatched line. This degradation converts fentanyl into norfentanyl ("NOR"), which is not an opioid, just a fragment of one. A second degradation, which is called amide hydrolysis, is a slower process (B) in which the hatched bond shown in blue is broken. This metabolism converts fentanyl into despropionylfentanyl ("DES"), which, like NOR is devoid of opioid activity. So, if fentanyl, NOR, and DES are all detected in the blood this is solid evidence that fentanyl was taken, but it does not tell us whether the drug was pharmaceutical or illicit. Except when it does.
In order to make this distinction we need a little more chemistry. (Sorry, no other way to do this). It turns out that DES is not only a minor metabolite of fentanyl but also, the synthetic precursor of the drug (1). All that is required is a very common chemical called propionyl chloride to easily convert DES into fentanyl (orange arrow) (2). If this reaction isn't properly performed, or the crude fentanyl product not purified, then there will very likely be some unknown amount of unreacted DES remaining as an impurity. It will be injected along with the fentanyl. A blood sample which shows high levels of DES suggests that the fentanyl in question came from an illicit lab because when pure fentanyl is metabolized very little DES if formed.
But in other instances, matters are far more definitive. There are about a dozen fentanyl analogs circulating around the US. Of these, only fentanyl can be legally used in people. So, when acetylfentanyl (an illegal analog) is used instead, a very different metabolic profile emerges (Figure 2).
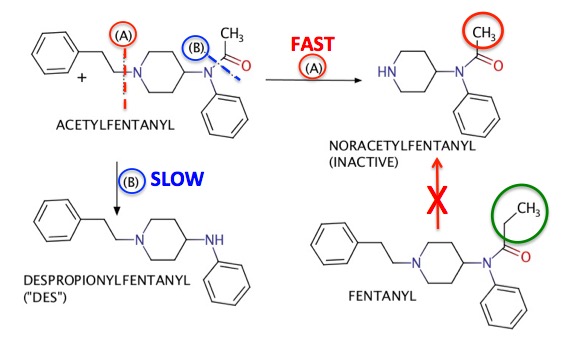
Figure 2. The metabolism of acetylfentanyl. As is the case with fentanyl, there is a fast metabolic step (red) and a slow one (blue). But when acetylfentanyl is the drug, the fast step forms noracetylfentanyl instead of norfentanyl - the known metabolite of fentanyl (Figure 1). This is where the difference lies. Since it is chemically and biochemically impossible (red arrow with X) to convert an ethyl group (green circle) into a methyl group (red circle) than the detection of noracetylfentanyl in blood is de facto evidence that the drug that was used was not from pharmaceutical fentanyl, but was instead illicit acetylfentanyl (3).
In reality, this type of analysis is more complicated because there are far more than two metabolites that form from these drugs, and there are a dozen different illicit fentanyl analogs to consider. But the concept is identical. By analyzing the chemicals of metabolism in the blood it is easy to determine the source of the drug. Probably easier than this chemistry lesson.
NOTES:
(1) This is only partly true. A knowledge of organic synthesis gives a chemist the ability to make a particular molecule or drug by a number of methods. There are a number of ways to make fentanyl. I am only showing the easiest. In fact, the green arrow represents another way to make fentanyl from NOR and a common chemical reagent called phenethyl bromide.
(2) Although DES is pharmacologically inactive it is still a Schedule II substance because of the ease of converting it to fentanyl.
(3) If acetylfentanyl is used it will, just like fentanyl it will also form a small amount of DES (blue), again by amide hydrolysis. But the presence of noracetylfentanyl precludes the possibility that the DES arose from fentanyl.




