It's not news that President Trump is having discussions with China's President Xi about getting a handle on fentanyl and "fentanyl-related substances." What is not widely known is that while an agreement between the two countries may be of some help in curbing the surge of opioid overdose deaths that we read about daily (1) there is a power that neither superpower can match - organic synthesis.
Organic synthesis is the science (and also art) of converting one molecule to another by using specific chemical reactions to bring about the desired transformation. Fentanyl itself is the product of organic synthesis; it was first made by chemist Paul Jannsen in 1960. Jannsen would become famous for forming Jannsen Pharmaceuticals, which is now part of Johnson and Johnson. Fentanyl would become famous for very different reasons.
Just as organic synthesis was responsible for the discovery of fentanyl it is also responsible for the "fentanyl-related substances," some of which are far more powerful than fentanyl itself, that are increasingly appearing in the US and being seized by the DEA. And while it may be possible to gain some control over fentanyl by legal means, "fentanyl-related substances" present a different (and worse) set of problems. And it's all because of organic chemistry.
The problem with the working plans is the term "fentanyl-related substances," which is another way of saying fentanyl analogs (2). There is no way to objectively define what this term means. Does it mean drugs that are structurally similar to fentanyl? Or does it mean drugs with the properties of fentanyl that can be put on a list and declared to be illegal? The first definition is subjective and will inevitably give wrong answers. The second is impossible. To make sense of any of this we need to take a look at the structure of fentanyl.
Two different chemists can look at the structure of just about any molecule and subdivide it into regions that he/she believes represents a "map" of the molecule; this is quite subjective. In Figure 1 I've shown two of an infinite number of possible "maps." Both structures are the fentanyl molecule and each contains four structural elements (again, arbitrary), but the structural elements are clearly different. Let's stick with the one on the left. It's simpler.
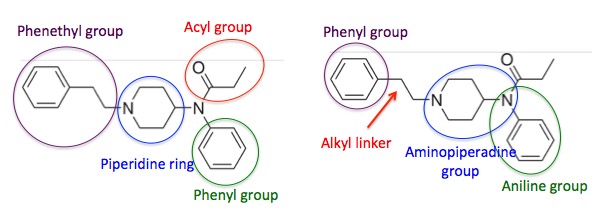
Figure 1. Two (of an infinite number) of different ways to look at the chemical structure of fentanyl, both arbitrary. The four colors represent "sections" of the fentanyl molecule. Note that the only difference is the placement of the circle, which determines the "section."
CATEGORIZING FENTANYL-LIKE SUBSTANCES BY STRUCTURAL SIMILARITY
Everything that it takes to poke a hole in this plan can be found in Figure 2. Whether you're a chemist or not it's pretty clear that despropionylfentanyl aka 4-ANPP (3) (Right) looks a whole lot like fentanyl (Left). Let's say that the "eyeball chemical similarity" is ~90%. So, it is reasonable to assume that these two chemicals would both be on the same list. But in order for it to be correct, that list would have to be entitled "Chemicals that look alike but behave completely differently." Little is known about the pharmacology of despropionylfentanyl but it sure isn't fentanyl; 4-ANPP is listed as an "irritant" on the safety data sheet of Matix Scientific, a supplier of the chemical. It is not an opioid even though it may "look" like one.
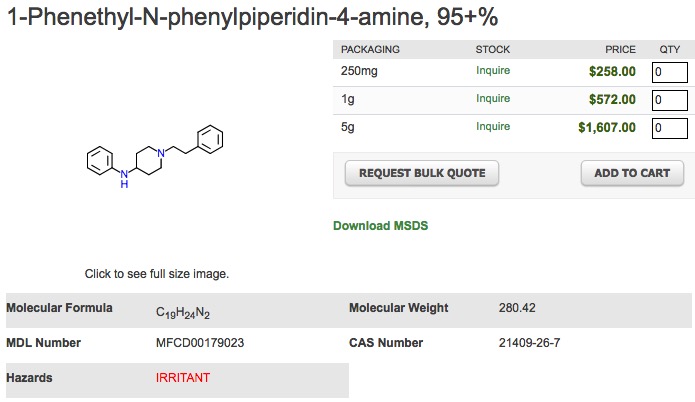
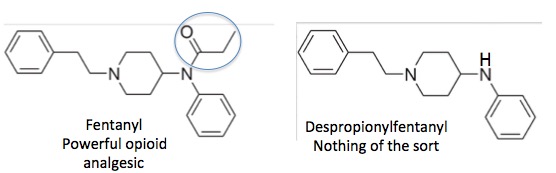
Figure 2. Fentanyl (Left) and despropionylfentanyl , aka 4-ANPP, (Right) look very similar but despropionylfentanyl has none of the properties of fentanyl. They differ only by the propionyl group (blue circle), but this makes all the difference in the world.
MAKING A LIST AND CHECKING IT A BILLION TIMES
There is no conceivable way of anticipating and listing all possible fentanyl analogs; the number is infinite. The DEA says that there are about 12 known fentanyl analogs that are now being circulated in the US. This does not even begin to scratch the surface of what could be synthesized. The vast majority of fentanyl analogs that we will see in the future don't even exist yet, except perhaps in the mind of a chemist. Or maybe he/she hasn't even thought of it yet.
A way to demonstrate the magnitude of the numbers is to look at one of several synthetic steps used to put fentanyl together. It is called acylation - the addition of an acetyl group to a molecule. When the molecule to be acylated contains an amine an amide bond is formed. Without this bond, the molecule will have no opioid properties.
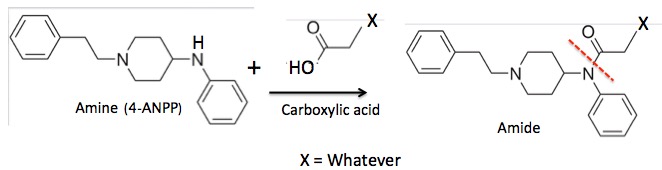
Figure 3. The reaction of 4-ANPP plus a carboxylic acid to form a fentanyl analog. The red hatch line indicates the critical amide bond. There are a huge number of possibilities for X, each yielding a different analog.
Here's one of many reasons why a list is not possible. If search the database of Sigma-Aldrich, the largest supplier of research chemicals, using "carboxylic acid" as the search term you'll see this:
The company sells 8,313 different carboxylic acids, so one could theoretically make 8,313 possible fentanyl analogs just from 4-ANPP (4). But it doesn't stop here. Not even close. Because fentanyl analogs don't need the exact 4-ANPP portion (also called the skeleton) of the molecule to be opioids. There are plenty of known fentanyl analogs that do not. Some look very much like fentanyl...
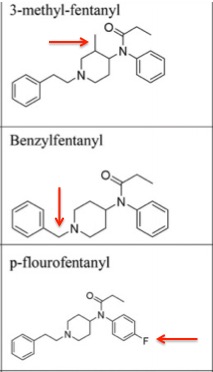
Figure 4. Close analogs of fentanyl. The red arrow shows a single change in the molecule. The analog at the top, 3-methylfentanyl is about 50-times more potent than fentanyl itself. Source: Jannetto, P. J. et. al., (2018). The Fentanyl Epidemic and Evolution of Fentanyl Analogs in the United States and the European Union. Clinical Chemistry, clinchem.2017.281626. doi:10.1373/clinchem.2017.281626
and some do not...
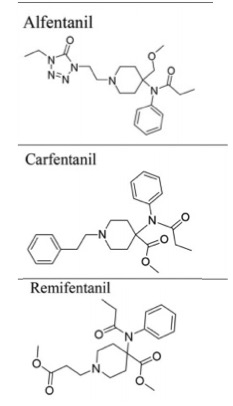
Figure 5. Analogs of fentanyl that are structurally dissimilar to fentanyl.
The take-home message here is that there are millions of fentanyl analogs that can be made from commercially available chemicals. It is all but certain that many of them will have fentanyl-like properties and that some of these will make even the most powerful analogs today, such as carfentanil and sufentanil, seem like cotton candy. The term used to describe the use of synthetic organic chemistry to maximize potency (and other properties) of potential drugs is "structure-activity relationships. (SAR)" It is the cornerstone of new drug discovery, both legal and otherwise. Conceptually it's very simple; SAR makes better analogs.
One tool that has been used by law-enforcement to ban new drugs that aren't specifically illegal is the 1986 Federal Analogue Act, which expands the definition of a controlled substance (5). The same language could be applied to an international agreement. Here is part of it:
"...the chemical structure of which is substantially similar to the chemical structure of a controlled substance..."
Here we go again. What is "substantially similar?" That depends on who you ask. It's just about impossible to come up with any kind of list or law that cannot be beaten by the power of organic synthesis, Figures 4 and 5 show us this.
And to make matters worse, where do you put this?
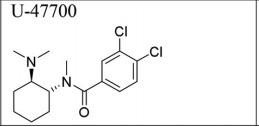
U-47700 is a potent opioid analgesic which is about seven-times stronger than morphine (6). It bears no resemblance to fentanyl but acts somewhat like it.
Good luck trying to fit U-47700 (and other analogs like it) onto a list of "fentanyl-related substances." Because it doesn't belong there, even though it's way more potent than morphine.
It should now be obvious that it is very far from simple to ban fentanyl and "fentanyl-related substances." Agreement with China or not, this mess isn't going away anytime soon. Chemists will see to that.
If you liked this piece, please consider donating to support our work. Happy Holidays!
NOTES:
(1) As I've written many times before, it is illicit fentanyl and its analogs that now are killing thousands of Americans every year, not Vicodin.
(2) Analogs are chemical cousins of a drug (or another chemical). They usually have a similar chemical structure and elicit a similar biological response, but both can also be quite different. The term is sometimes subjective.
(3) Despropionylfentanyl, 4-Aminophenyl-1-phenethylpiperidine, and 4-ANPP are all names for the same molecule. It takes about 2 seconds to convert it to fentanyl, so it is highly controlled, so you'll never be able to buy it. But a competent organic chemist can just make it from any number of smaller, common chemicals. It just takes a bit more work. See how easy this is?
(4) No one is right or wrong mind would ever try to make all of these. Most of the "fentanyl derivatives" made from complex or large carboxylic acids wouldn't work. But there are probably a couple of hundred that would. Organic chemists just know this stuff.
(5) The act makes it possible to declare drugs illegal, even though they are not even known yet. Unless you get a good lawyer who can convince the court that whatever you're caught with isn't "similar enough."
(6) Reference: Cheney BV, Szmuszkovicz J, Lahti RA, Zichi DA (December 1985). "Factors affecting binding of trans-N-[2-(methylamino)cyclohexyl]benzamides at the primary morphine receptor". Journal of Medicinal Chemistry. 28 (12): 1853–64.