By Dr. Michael Dourson and Dr. Bernard Gadagbui
A recent meeting of the Society of Environmental Toxicology and Chemistry (SETAC) demonstrated that government agencies throughout the world are facing a dilemma: a wide divergence on the safety assessment of these chemistries, thus, making communication with the various publics extremely difficult, if not impossible.
This dilemma appears to be related to the one clear message from this SETAC meeting: the assessment of perfluorinated chemicals is extraordinarily complex. This message reverberated in each of the various sessions of the 2019 SETAC meeting: Environmental Sources, Chemistry, Fate and Transport; Exposure Assessment; Ecological Toxicity; Human Health Toxicity; and Risk Characterization.
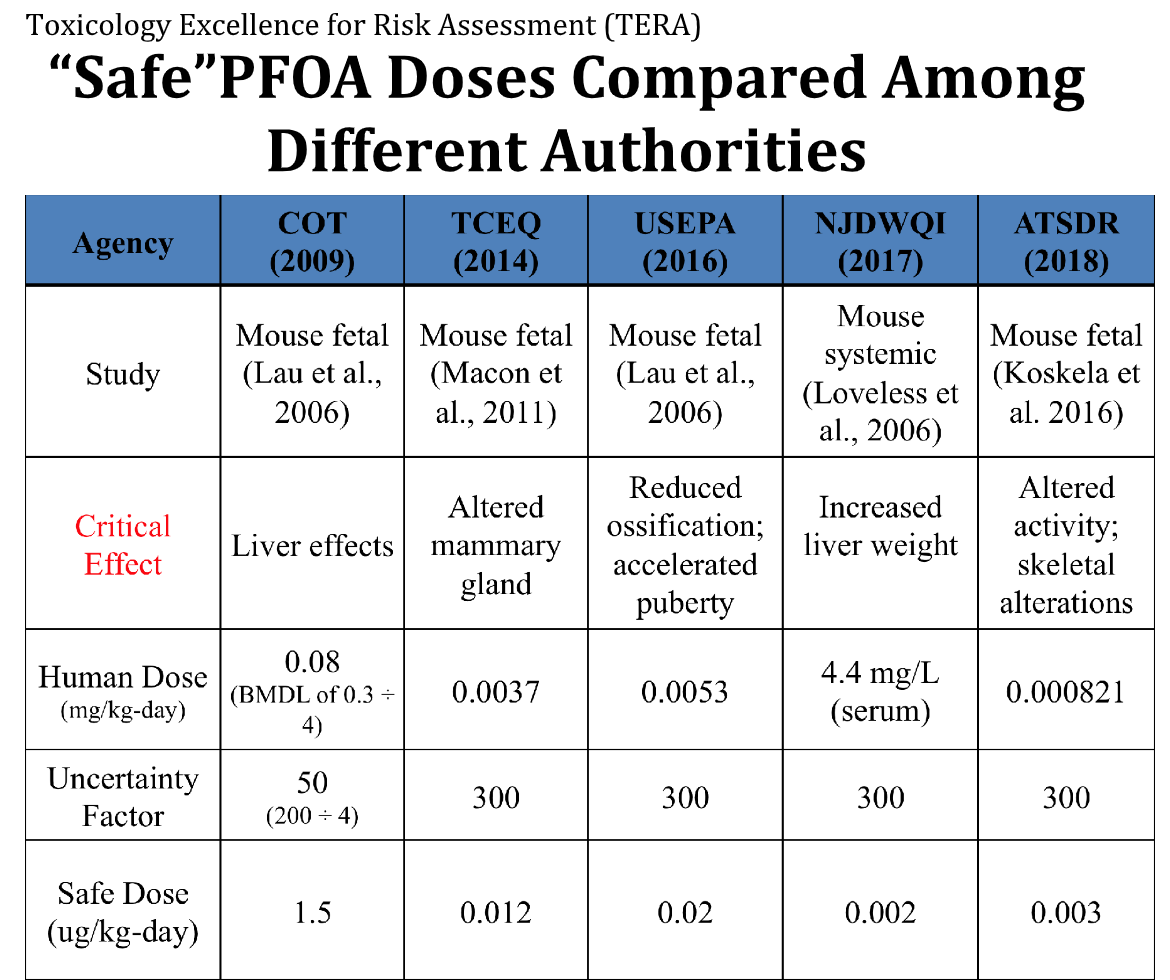 To cite just one example, the chemical perfluorooctanoate (PFOA) has estimates of its safe dose by government agencies that vary by around 750-fold. (See chart.) Scientists within these various agencies are well informed and quite experienced. So how is it then that such diversity can exist in the estimation of the PFOA safe dose?
To cite just one example, the chemical perfluorooctanoate (PFOA) has estimates of its safe dose by government agencies that vary by around 750-fold. (See chart.) Scientists within these various agencies are well informed and quite experienced. So how is it then that such diversity can exist in the estimation of the PFOA safe dose?
In part, this diversity of opinion is because the underlying science in determination of PFOA's first toxic effect is not straightforward, and government agencies do not agree, as explained in an earlier post by Dr. Jenifer Health.
Furthermore, as she explains, the extrapolation from experimental animals to humans involves understanding of how long these chemicals stay in our bodies (referred to as half-life). Humans appear to have a longer half-life (measured in years) than the half-life (measured in days) of the experimental animals used in the estimation of these various safe doses (the larger the difference in half-life, the lower the safe dose is in humans). However, not all agencies agree with this difference in half-life either, mainly because the estimates of half-life in humans are generally based on observational studies in humans, where not all sources of PFOA exposure may have been carefully monitored1. Both disagreements lead to the wide divergence in PFOA safe doses.
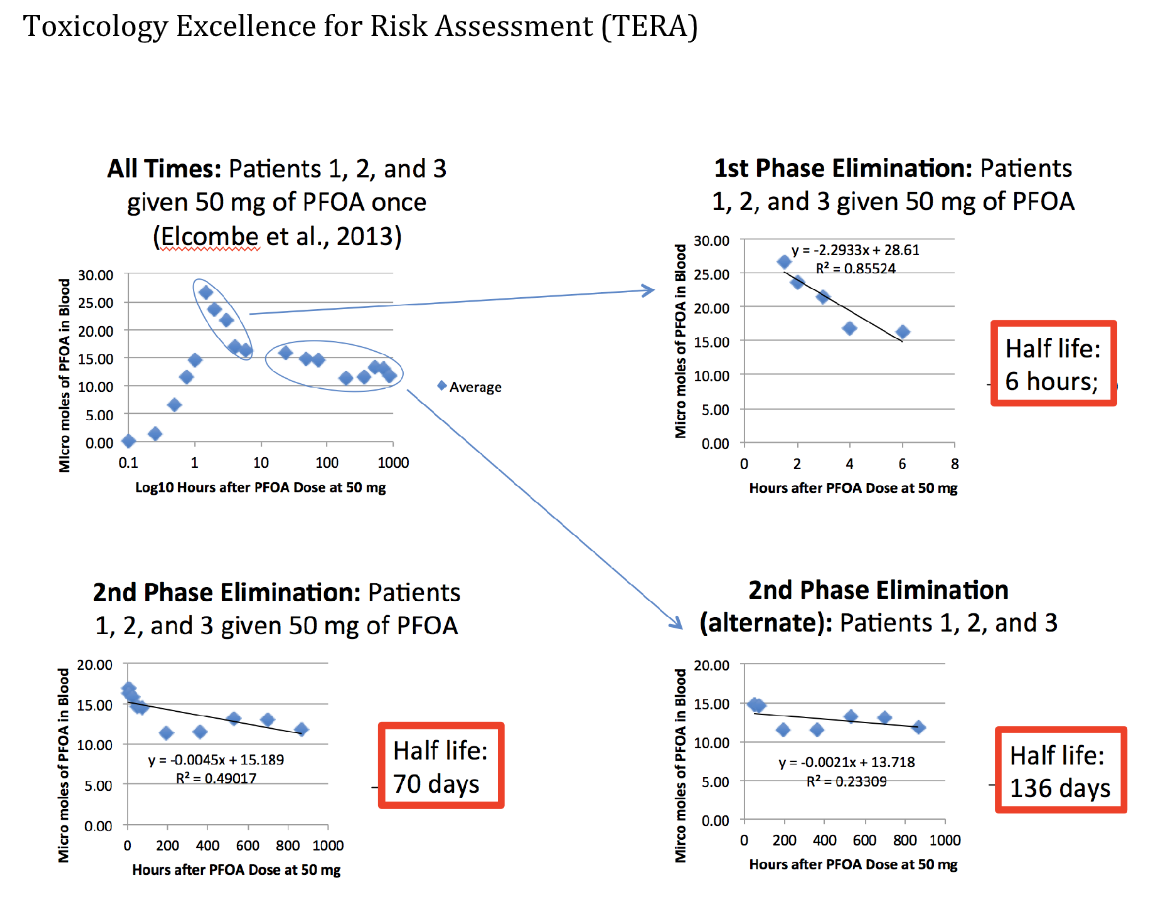 Fortuitously, a recently available human study by Elcombe et al. (2013) has been analyzed2 that may help resolve in part this dilemma in half-life. Forty-three patients in various stages of cancer were given PFOA as part of a clinical trial. Adequate liver and kidney function were a requirement for participating. PFOA concentrations in blood were carefully followed. Three of these patients were only given one dose. The blood concentration of PFOA in these three patients over 6 weeks suggests a two-phase half-life, with the second phase, between 70 and 140 days, that is very different from the half-life estimated in years based on the human observational studies as shown in the second chart.
Fortuitously, a recently available human study by Elcombe et al. (2013) has been analyzed2 that may help resolve in part this dilemma in half-life. Forty-three patients in various stages of cancer were given PFOA as part of a clinical trial. Adequate liver and kidney function were a requirement for participating. PFOA concentrations in blood were carefully followed. Three of these patients were only given one dose. The blood concentration of PFOA in these three patients over 6 weeks suggests a two-phase half-life, with the second phase, between 70 and 140 days, that is very different from the half-life estimated in years based on the human observational studies as shown in the second chart.
Of course, this latter estimated range is preliminary and based on only three humans who were sick, and so the half-life in these patients may have been shortened. Alternatively, PFOA may have a slower third phase in its half-life that may be operating beyond six weeks. Not surprisingly, an ongoing effort is now being conducted to integrate the findings from all 43 patients into a half-life estimate, and then compare this to half-life estimates from the human observational studies. Hopefully, this will resolve part of the dilemma now facing government agencies world-wide.
Stay tuned!
Footnotes
(1) The SETAC conference made it clear that PFOA and other PFAS chemicals are now known to be pervasive. Thus, older human intervention studies that did not actively monitor other sources of exposure, such as fabric, household dust, food and/or air, will likely have half-life estimates that are too large.
(2) Dourson et al. (2019) partially analyzes this study, which is now available on line and with free access at https://authors.elsevier.com/a/1Zbd~~81IFXBR until October 9, 2019. Research highlights:
- The choice of the appropriate dosimeter is important in the development of a Data Derived Extrapolation Factor (DDEF).
- Research shows derivation of a DDEF for developmental toxicity for PFOA.
- Comparison made of kinetic data from PFOA exposure in mice with carefully monitored kinetic data in humans.
- A DDEF for PFOA was determined to be 1.3 or 14 depending on choice of critical effect.
References
ATSDR (Agency for Toxic Substances and Disease Registry), 2018. Toxicological Profile for Perfluoroalkyls Draft for Public Comment. U.S. Department of Health and Human Services. https://www.atsdr.cdc.gov/toxprofiles/tp200.pdf.
Committee on Toxicity, 2009. Update Statement on the Tolerable Daily Intake for Perfluorooctanoic acid. Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment. United Kingdom. https://cot.food.gov.uk/sites/default/files/cot/cotstatementpfoa200902.pdf.
Dourson M.L., B. Gadagbui, C. Onyema, P.M. McGinnis and R.G. York. 2019. Data Derived Extrapolation Factors for Developmental Toxicity: A Preliminary Research Case Study with Perfluorooctanoate (PFOA). Regulatory Toxicology and Pharmacology. Volume 108, November 2019, 104446.
Elcombe, C.R., Wolf, C.R., Westwood, A.L., 2013. US Patent Application Publication, Pub. No.: US 2013/0029928. Available at: https://patentimages.storage.googleapis.com/24/ee/73/f58267c7d70dde/WO20....
EPA (United States Environmental Protection Agency), 2016. Health Effects Support Document for Perfluorooctanoic Acid (PFOA). Office of Water (4304T) Health and Ecological Criteria Division Washington, DC 20460. EPA 822-R-16-003. May 2016. https://www.epa.gov/sites/production/files/2016-05/documents/pfoa_hesd_final-plain.pdf.
NJDWQI (New Jersey Drinking Water Quality Institute), 2017. Health-based Maximum Contaminant Level Support Document: Perfluorooctanoic Acid (PFOA). https://www.nj.gov/dep/watersupply/pdf/pfoa-appendixa.pdf.
TCEQ (Texas Commission on Environmental Quality), 2016. Perfluoro Compounds (PFCs) Various CASRN Numbers. January 4. https://www.tceq.texas.gov/assets/public/implementation/tox/evaluations/pfcs.pdf.




