ProPublica has made uncovering the financial conflicts of interest between prescribers and pharmaceutical and medical device companies a bit of a cottage industry. Their Dollars for Docs (they couldn’t even complete the word, we are doctors) is fueled by required disclosures by companies “under the Physician Payments Sunshine Act, a part of the 2010 Affordable Care Act.”
Often the cited numbers lack nuance and context. For example, in 2018, Dr. Kevin Foley, a neurosurgeon, collected almost $29 million from these companies. But a “rounding error,” $30,000 was for consultation and speaking; all the rest were royalties and licensure of his inventions. But I digress. The current research attempted to describe a healthcare “ecosystem” rife with industry influence.
A tangled web
“Central to the ecosystem are healthcare providers, researchers, clinical care facilities, journals, professional societies, and other healthcare institutions and supporting organizations engaged in medicine’s core professional activities: providing beneficial care to patients, conducting valid research, and providing evidence based clinical education and guidance.”
Let’s see the map of the influencers where a picture is worth at least 1000 words.
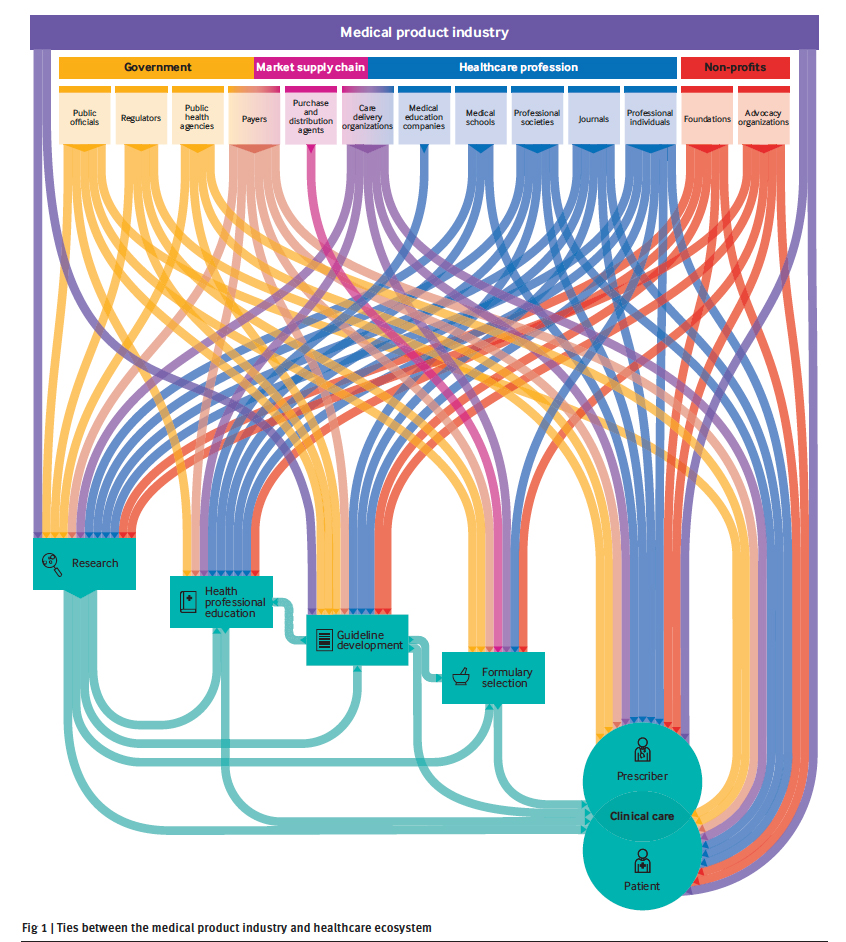
This mapping was based on a literature review and interviews with “an international panel of experts with broad expertise in industry ties and deep knowledge of specific parties and activities.” Prescribers are a subset of professional individuals; they are on the front lines prescribing medications and using devices. Professional individuals include those involved in research, the drafting of guidelines, professional education, and selection of drugs to be used (formulary selection)
Everyone is connected, and as they report,
“All party types have financial ties to medical product companies” [emphasis added]
Except for payers and distributors, all of the parties have additional non-financial ties. You can’t help but bump into a conflict of interest if that is the lens you choose to use.
The government and product manufacturers fund all five areas of medical concern: research, professional education, guideline development, formulary selection [1], and clinical care involving the interaction of patients and healthcare professionals – “Ask your doctor about” (fill in with any drug or disease).
ProPublica’s database and the Sunshine Law both look at the final critical interaction between patient and prescriber. Still, as the researchers point out, that moment “is shaped by all other activities and is the ultimate target of industry interest.” But in our haste to focus on the final interaction, those other conflicted moments have not received as much attention. For example, 56% of literature citations deal with industry and research, 29% deal with industry ties to clinical care, and 27% to industry ties to professional education. Guideline development and formulary selection, points where standards are set, have only interested researchers 6% of the time, and only eight reviewed publications (1%) considered industry relationships with journals.
More importantly, especially from the academic perspective, while financial ties are almost always subject to policy and oversight, the same is not true for non-financial ties. Working on drug or device research generates peer-review papers, the coin of the realm in academic advancement. Industry connections also raise academicians' stature so that they sit on guideline committees or advisory boards to the government, and yes, to industry. The role of patient advocacy groups is also a bit uncharted. Some have industry funding, and many expensive medications have co-payment arrangements that shift costs away from patients and onto their health plans.
One of the vital points made by the researchers is the “outsized focus” on payments to prescribers. In part, this is because the information is more transparently available. But as they write,
“By compiling and mapping the full network of the medical product industry’s reach across the healthcare ecosystem, we depict the ways in which potential influence moves well beyond the spheres of individual professionals and prescribers.”
Limitations
The researchers mention three. There may be more hidden connections they have not identified. I would suggest that there may be a bit of bias here because they fail to discuss the impacts of funding upon academic tenure and standing. They also cannot measure the magnitude of these conflicts, and more importantly, they only identify potential conflicts.
Conflicts of interest are found in our hearts, not necessarily in our behavior. One of the subtle phrasings in the article is that influence moves in one direction.
“The pharmaceutical and medical device and biotechnology industries have established numerous ties with non-profit entities, the healthcare profession, the market supply chain, and government.”
Influence moves in both directions. Health systems, professionals, supply chain intermediaries, and the government are all reaching out to Big Pharma and Big Device.
Bonus
One of the supplements provided by the researchers discusses the role of influencers in the “opioid” crisis. ACSH and especially Dr. Bloom have written extensively on how the opioid crisis is not driven by responsible physician prescribing and how the CDC guidelines have hurt more than helped. You can access the supplement here. It is not exhaustive and only reflects the mainstream narrative of prescription medications being the underlying driver – that said, it shows the breadth of conflicts when you follow the money.
[1] Formulary selection is an unsung but vital roadblock. You cannot prescribe a drug as a physician, or it costs the patients significantly more in out-of-pocket costs to use a medication, not on formulary in a health plan or system. The battles over what gets approved for general use and what is limited to approval by specialists can be very heated.
Source: Mapping Conflict of Interests BMJ DOI: 10.1136/bmj-2021-066576




