A recent article in the New York Times discussed potential risks from some of the inks (and other ingredients) used to create tattoos, which now festoon the skin of about 12% of people in the EU, where two dyes (colorants) will be banned due to health concerns, real or imagined. This brings up a couple of obvious questions:
- Are there toxic chemicals in tattoo ink? Yes. (1)
- Do they cause harm? Since harm involves both the inherent toxicity of a chemical and the dose (in this case, the amount of ink), there is no easy way to answer this question.
EU to the rescue
These concerns prompted the European Chemicals Agency (ECHA), a public health agency of the EU, to evaluate the potential risks of inks and other chemicals used for tattooing, especially those that are known to be toxic, mutagenic, or carcinogenic. After all, since they are injected into the skin where they can stay for life, some of these chemicals will migrate to the lymph nodes and liver – not exactly the places where you'd want carcinogens to collect. As a result, in early 2022, the EU banned thousands of chemicals that were previously used for tattoos and permanent makeup.
Although the EU is notoriously chemophobic (sometimes ridiculously so), the study of tattoos has also spurred interest in the US. Studies of the composition and safety of inks and their carriers – the chemicals used to deposit them into the skin – were one topic at the American Chemical Society's Fall Meeting 2022 this past August.
Quality control or the lack thereof
John Swierk, Ph.D, a chemistry professor at SUNY Binghamton, and his group has been investigating what's actually in the inks as well as what happens when it's removed from the skin. Swierk presented his results at the ACS meeting:
“The idea for this project initially came about because I was interested in what happens when laser light is used to remove tattoos...But then I realized that very little is actually known about the composition of tattoo inks, so we started analyzing popular brands.”'
John Swierk, Ph.D
Swierk analyzed 100 inks. He found that they didn't exactly come flying through quality control with colors (yes - pun):
“Every time we looked at one of the inks, we found something that gave me pause...For example, 23 of 56 different inks analyzed to date suggest an azo-containing dye is present.” (2)
Singing the blues for blue and green
Of the thousands of chemicals banned by the EU, two are of particular interest. Blue 15:3 and Green 7 are present in 70% of tattoos, and both found themselves on the list, something that was deeply troubling to ink manufacturers because there are no suitable replacements.
The ban was not well received by the tattoo industry, as evidenced by a comical-sounding website in Australia: Save the inks! Early reports suggest that whales are not happy with this name; however, no comment could be obtained from the "Whales of the World" spokes mammal as he was busy trying to dodge Japanese hunting ships.
Why are these colors different?
Why don't people who are concerned about the health effects of inks etc. just have their tattoos removed? Not so simple. The conditions used to remove the tattoos can break down the chemicals in some inks and then release a different set of potentially hazardous substances.
Because of the importance and wide use of blue and green inks, the laser-induced decomposition of these chemicals has been studied. A 2015 paper in Nature Scientific Reports examined what happened when Blue 15:3 and Green 7 (3,4) were subjected to heat and radiation from a laser.
"Since laser treatment of tattoos is the favored method for the removing of no longer wanted permanent skin paintings, analytical, biokinetics and toxicological data on the fragmentation pattern of commonly used pigments are urgently required for health safety reasons."
Schreiver, I., Hutzler, C., Laux, P. et al. Formation of highly toxic hydrogen cyanide upon ruby laser irradiation of the tattoo pigment phthalocyanine blue. Sci Rep 5, 12915 (2015). https://doi.org/10.1038/srep12915
The results were quite interesting (Figure 1). Non-chemists may want to look away...
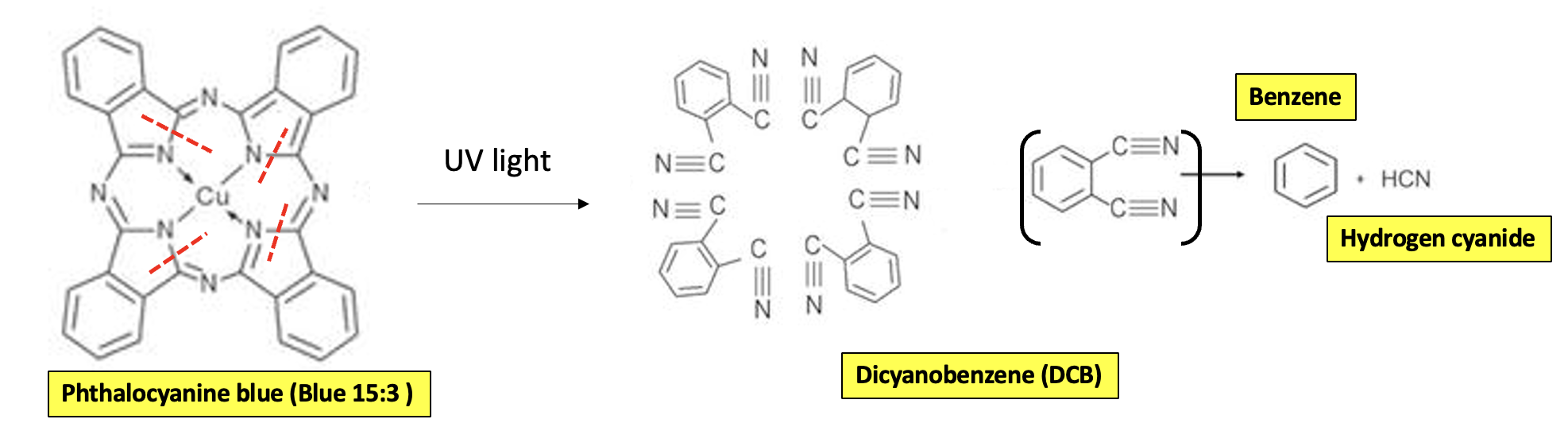
Figure 1. Decomposition pattern of Phthalocyanine Blue (PB15:3) based on the pyrolysis and laser irradiation. Blue 15:3, aka copper phthalocyanine, absorbs light and decomposes to give 4 molecules of dicyanobenzene (DCB). DCB can further react, losing hydrogen cyanide (toxic) and forming benzene - a known carcinogen. The red hatched lines indicate the bonds that are broken by the irradiation. Source: Nature Scientific Reports
Bottom line
Do tattoos and/or their removal present a real health threat to the inked of the world? It's hard to say. There are certainly theoretical risks that, at least chemically, make sense, but a consensus seems to be that there is little to worry about when proper precaution is taken:
In general, when proper precautions are taken, tattoos are unlikely to cause major health problems. The most common complications associated with tattoos are skin infections, especially staphylococcus aureus and streptococcus pyogenes, allergic contact dermatitis and granulomas (inflammation that causes small bumps in the skin). Viral infections, such as HIV and hepatitis from contaminated needles, are extremely rare but still possible.
Source: Cancer Treatment Centers of America
One exception
Although no thorough data exists, anecdotal evidence suggests that real and immediate danger is possible if you have "Yankees" tattooed on your forehead on a visit to Fenway Park. But this is an unlikely scenario. Those Yankee fans who have not (yet) hung themselves in the middle of an ongoing epic collapse are allegedly having such tattoos removed in record numbers.
NOTES:
(1) There is not a chemical on earth that is not toxic at a given dose. Nor is there one that is non-toxic at a different (and much lower) dose.
(2) "All azo pigments are in general prohibited in tattoo dyes if their reductive azo cleavage could lead to primary arylamines with carcinogenic, mutagenic, or reprotoxic characteristics." Source: Chemistry Views
(3) I have not discussed Green 7 because it is an analog of Blue 15:3 with all the hydrogen atoms replaced by chlorine. The chemistry of decomposition is the same.
(4) Here's a book chapter that gives a comprehensive review of the chemistry of the breakdown of tattoo ink. See Photochemical Degradation Processes of Painting Materials from Cultural Heritage. Don't expect easy reading.




