Dear Garlic Aficionados,
Have you (or someone downwind of you) ever noticed that your breath really begins to stink well after a garlicesque (welcome to National Make Up Stupid Words Day) meal, perhaps even the next day? Or that the stench that ultimately spews forth from your body doesn’t really smell like yummy garlic anymore; it’s a noticeably different, pungent, unpleasant odor. It all makes sense, though. This is how your body handles garlic, something you will find out shortly.
Naturally, garlic eaters, especially those subsequently locked out of their homes by anti-garlites, will attempt to degarlicize themselves by using mouthwash. But it won’t work. It can’t. Mysterious, no?
Actually, not at all. The simple answer to this delectable puzzle is how your body metabolizes the many sulfur-containing chemicals in garlic and what happens to them afterward. It makes its way from the blood to the lungs and is expelled with every breath, regardless of whether this occurs via your mouth or nose. This is why mouthwash won’t make a damn bit of difference. The smell is systemic; whatever chemical is responsible comes from your lungs – not your mouth. Tiny amounts of it can even be expelled through your skin. So you might as well try nose gargling for all the good that mouthwash is gonna do.
The good news is that by the time you finish reading this, you will still stink but will have saved from gargling with Listerine, a vile concoction that tastes (just my opinion) like an equimolar mixture of battery acid and camel pee.
To explain what’s going on, we need a long overdue episode of…

Steve (left) and Irwin, your perpetually cantankerous hosts of TDCLFH®, delicately try to navigate the perpetual landmine of interpersonal matters that arise from being roommates for more than 1,000 years.
Here's a rule that organic chemists know all too well. Organosulfur compounds stink. Pretty much any volatile, sulfur-containing chemical is going to smell really bad. A few (of many) examples:
- hydrogen sulfide (rotten eggs)
- isoamyl mercaptan (skunk)
- methanethiol (farts)
- dimethyl sulfide (cabbage and the ocean)
- t-butyl mercaptan (odorant in cooking gases)
Garlic is the poster child for sulfur promiscuity. It contains a whole bunch (dozens) of sulfur-containing chemicals. Here are some of them. There is a reason I put that mess below in front of you, even though you may prefer to brush your teeth with a chainsaw than look at it.
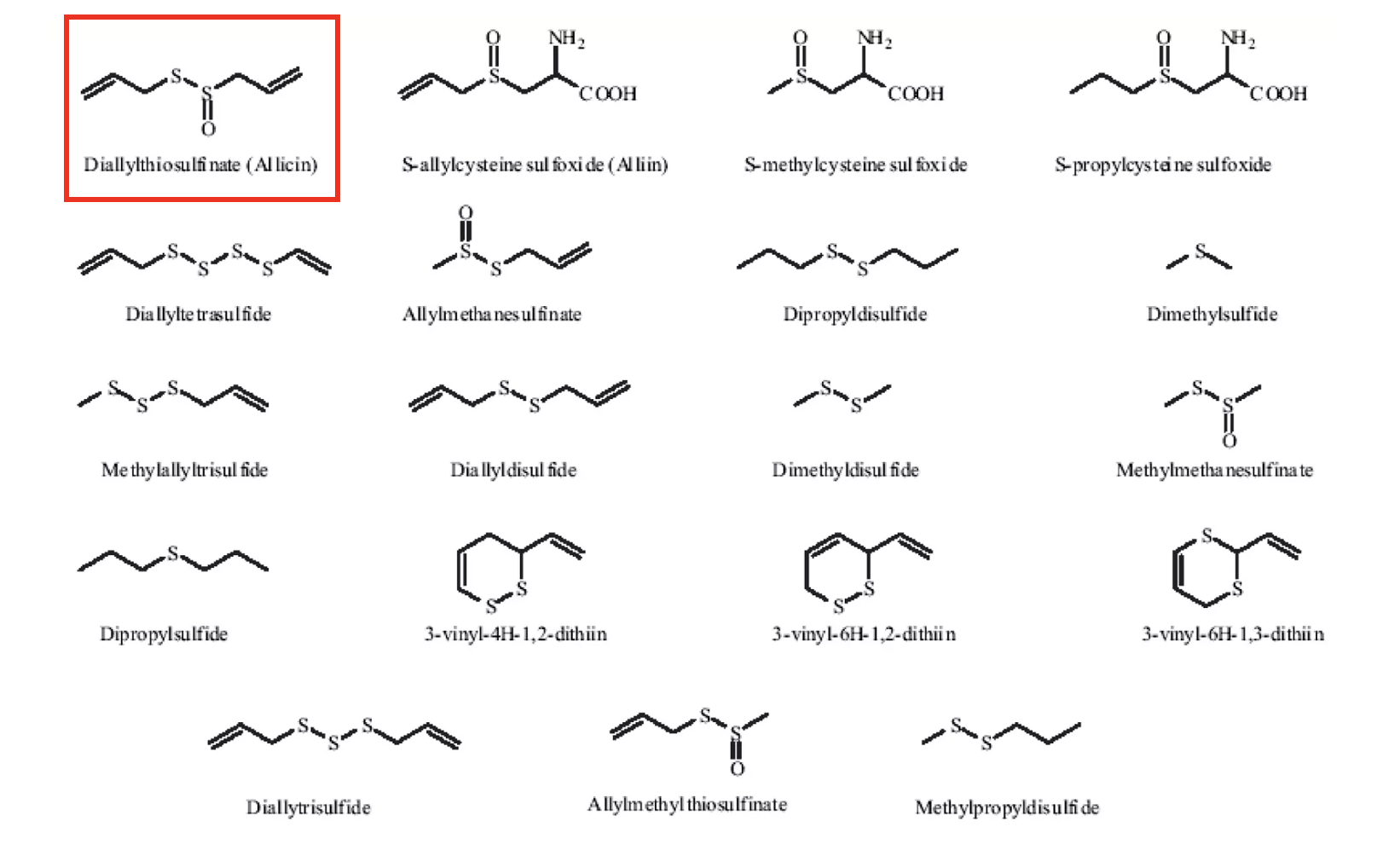
Figure 1. A whole bunch of garlic chemicals that you have absolutely no interest in seeing. Except for one in the red box. And then, only maybe. Source: Slide Share
The principal odor chemical in garlic is a chemical called allicin (red box). Look at it. Whatever attributes allicin has going for it, stability isn't one of them. By the time it has reached your stomach, it is oozing its way toward its demise. Whatever little manages to get as far as the liver need not send a letter with a return address label. It won't be around long.
As shown in Figure 2, first, the sulfur-sulfur bond is broken, releasing a hideous stench monster called allyl mercaptan, which is methylated by a ubiquitous enzyme called S-adenosylmethionine to form allyl methyl sulfide – the chemical that has you locked out of your house. Note the difference in volatility. Allicin boils at 249 +/- 43 (huh??) oC, or 480 oF, while allyl methyl sulfide boils at 92oC, a temperature near that of alcohol and water. This is why it is so volatile. It is also stable, hence the stench.
(OK, WTF is going on here? 249 +/- 43oC?? That sounds like a pretty wide range. Sort of like the weather person on the news predicting: "Tomorrow will be partly cloudy, and the temperature will range from 34 to 85. Dress accordingly." The answer is that this is a calculated, not measured, value. Check out ChemSpider. You'll see this method used commonly.)
Now, go back to Figure 1, which, if you had any sense, you would have ignored in the first place. Look for allyl methyl sulfide. It ain’t there. The chemical coming from your lungs the next day isn’t even found in garlic. Otherwise, pre-digested garlic would smell a helluva lot different.
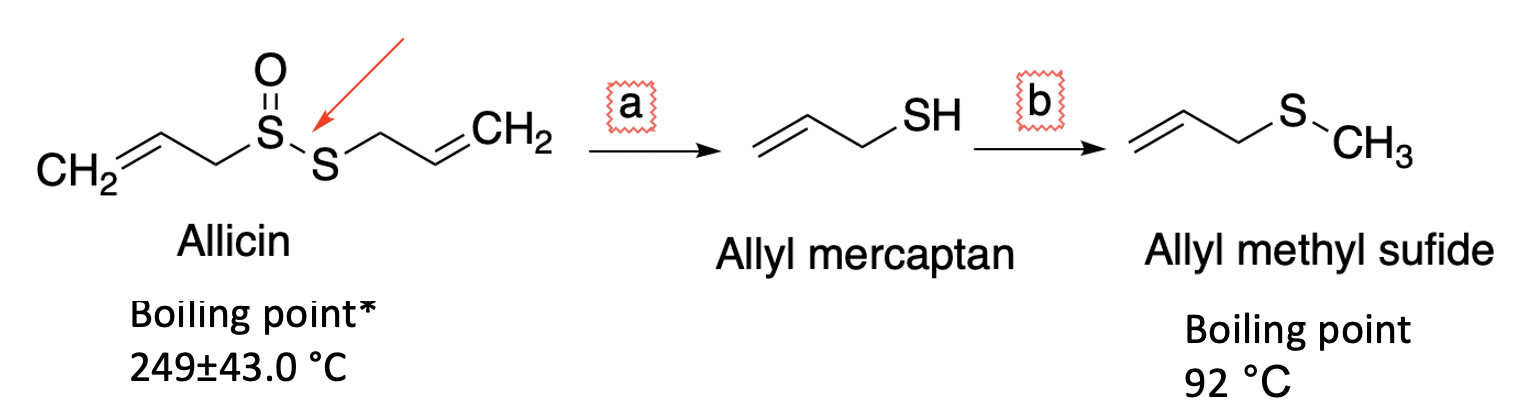
Figure 2. Human metabolism of allicin begins with splitting the sulfur-sulfur bond (a, red arrow) by a well-known process common to many disulfides and their derivatives. This forms allyl mercaptan, which undergoes methylation (b) by a common enzyme to form allyl methyl sulfide, which is stable. And stinky.
Is there a message here other than some mindless chemistry? Maybe. You might want to lay off the garlic if you have an online date the next day. Your already minuscule chances of meeting someone who isn't an axe murderer is a compatible life partner may improve a bit. I haven't seen a whole lot of Times wedding announcements where the couple met at a garlic festival.




