Sepsis is an overwhelming infection that can lead to organ failure and death. It is a big problem.
In the U.S., 1.6 million patients are affected annually, with about 250,000 dying – far greater than the deaths from breast cancer, which garner much more publicity. Moreover, sepsis requires rapid diagnosis and treatment making it a priority in our Emergency Departments. For the economically minded, it is the most expensive care we provide. [1]
An article in the American Chemical Society Infectious Disease suggests we have overlooked a means of treating sepsis that our body already provides, bicarbonate. It is important to note that the research involves in vitro work only.
Of Bicarbonate and Bacteria
Bicarbonate acts in conjunction with carbon dioxide to buffer our blood, maintaining our pH around 7.4 – i.e., slightly basic. Your kidneys control the bicarbonate level, holding on or releasing bicarbonate; your lungs control the carbon dioxide, increasing or decreasing your respiratory rate to blow off or retain carbon dioxide. Changes in the bicarbonate level of your blood occur more slowly than the respiratory changes that alter carbon dioxide levels.
Bacterial sensitivity to antibiotics is measured by culturing the bacteria on Petri dishes and plopping little discs with various antibiotics on the plates and seeing what doesn’t grow. The plates are bathed in solutions that mimic our physiology, with a "normal" level of bicarbonate and pH. Bacterial sensitivities to antibiotics are expressed as minimum inhibitory concentrations (MIC); i.e., the concentration of antibiotic that is necessary to stop bacterial growth. The authors noted that studies had shown both increased and decreased MICs when bicarbonate levels were altered. They asked whether bicarbonate, by itself, had antimicrobial effects.
Their experiments demonstrate that at physiologic concentrations, bicarbonate enhanced the activity of some antibiotics. More importantly, with respect to sepsis, bicarbonate potentiated the impact of antibiotics on four of the bacterial species identified by the World Health Organization as global health priorities. [2] This effect was not related to an increase in pH, which was adjusted to remain physiologically normal.
Their hypothesis for these findings was that bicarbonate interfered with the energy available to bacteria, by altering what is known as the proton motive force (PMF), an electrochemical gradient across the bacterial cell wall that bacteria harness to make energy. Bicarbonate interferes with this gradient, and this can influence how antibiotic molecules behave.
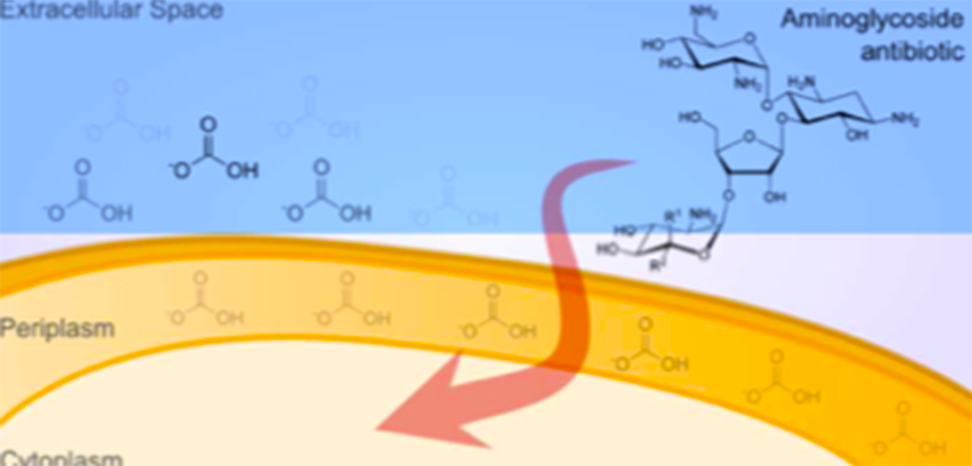
For instance, the PMF is directly related to pumps that bacteria possess in their cell membranes. Tetracycline penetrates cell walls based on a pH gradient, but bicarbonate reduces the gradient, reducing tetracycline's effect as measured by MICs. As bicarbonate reduces the pH gradient, bacteria compensate by increasing the electrical gradient. Aminoglycosides' activity is more related to electrical charge, and bicarbonate enhances the entry of aminoglycosides as measured by MICs.
Additionally, under normal physiologic concentrations, bicarbonate seems to make it more difficult for bacteria like E. coli to produce energy, resulting in slower growth. Slower growth means that antibiotics that act when bacteria are actively dividing become less effective. Furthermore, their research suggested that bicarbonate modulates portions of our immune response and can have impacts on antibiotics that we fail to recognize from simple in vitro testing.
The authors believe these experiments can direct research into new antimicrobial drugs and therapies. But as a clinician, I think the experimental findings point towards a more immediate use for bicarbonate, as well.
Could Bicarbonate Fight Sepsis?
Sepsis has multiple physiologic impacts, but a crucial event is an interference with the efficient use of oxygen by cells, switching increasingly to pathways that produce less energy and a build-up of metabolic acids. These acids lower the pH, and the body corrects for this by holding on to bicarbonate, a base, and increasing the respiratory rate, to blow off carbon dioxide, an acid. As sepsis intensifies, the kidneys cannot hold on to enough bicarbonate, and your respiratory rate cannot go fast enough to compensate adequately, and your pH becomes more acidic. [3]
Oxygen binds to the hemoglobin in our cells where it is carried by our circulation to the cells. In an acidic environment, oxygen binds more tightly to hemoglobin and less and less is released to the tissue that needs it for energy production. This, in turn, hastens the creation of more metabolic acids and a continued lowering of pH -- a feedback loop that spirals downward. Patients with severe sepsis are often placed on ventilators to control their oxygenation and breathing. Thus, doctors rather than the human body take over management of carbon dioxide and the provision of bicarbonate that the kidneys can maintain. Physicians are good, but not as good as a set of healthy lungs and kidneys.
The other consequence of the increasingly inefficient energy utilization is that our heart begins to fail. To maintain adequate flow, our blood vessels dilate so that the heart does not have to work as hard. (Technically, vessels dilate to lower the resistance for the work of the heart). Physicians can support these functions with drugs that increase the pumping ability of the heart and adjust the blood vessels to augment and optimize circulation. We know these drugs are less effective in acidic environments, so physicians will provide supplemental bicarbonate to improve the drug’s action and subsequently support the pumping of the heart.
There is an ongoing debate about the role of bicarbonate in sepsis. Current guidelines suggest clinicians supplement bicarbonate when the patient’s pH falls below 7.2. (Normal pH is around 7.4.) Our current clinical belief is that bicarbonate's role is in supporting circulation, so the timing of our intervention is based upon circulatory measures, like blood pressure. [4] The findings in this paper show that bicarbonate may have a greater role than merely supporting circulation.
What if providing bicarbonate was based on enhancing the response of antibiotics? What if an antibiotic selection was geared not only to the bacterial susceptibility but the patient’s pH? Is it possible that we could improve the outcome of sepsis, by supporting the body’s stores of bicarbonate earlier, guiding therapy to enhance antimicrobial efficacy? Is it possible that heart function and blood pressure are the wrong criteria for action? Are the failures of circulation a measurement too late in the process, that the death spiral is already underway?
We can study providing supplemental bicarbonate to septic patients now, and for me, that is the most significant promise of this paper. Bicarbonate may prove to be a simple means to improve care for a tough problem, with an inexpensive and readily available medication. Indeed, it's just baking soda.
Notes
[1] Sepsis Alliance Sepsis Fact Sheet
[2] Acinetobacter baumannii, Klebsiella Pneumoniae, Pseudomonas aeruginosa, and methicillin-resistant staph aureus.
[3] This is referred to as a respiratory alkalosis and combined metabolic alkalosis and acidosis.
[4] The current guidelines recommend against the use of bicarbonate to maintain circulation or enhance the effect of medications chosen to increase blood pressure and circulation until a pH of 7.15
Source: Bicarbonate alters bacterial susceptibility to antibiotics by targeting the proton motive force ACS Infectious Diseases, Article ASAP DOI: 10.1021/acsinfecdis.7b00194




