Expert societies, especially the infectious disease pharmacists, should consider making recommendations regarding off-label use of antibiotics. These recommendations should be based on clinical, microbiological, and pharmacodynamic/pharmacokinetic data.
I have a modest proposal that comes from my recent discussions with Brad Spellberg, Lew Barrett and John Rex. I am grateful to all three of these experts for their thoughts on this topic.
To successfully commercialize a new antibiotic, the drug must be shown in clinical trials and via microbiology and pharmacokinetic / pharmacodynamic (PK/PD) data to provide advantages that fill a medical need. An obvious example would be if someone markets an oral antibacterial active against multple drug-resistant (MDR) pathogens that works clinically in complicated urinary tract infection. The “educational” activities (marketing) required to convince physicians and pharmacists that any new agent should be available to physicians via their hospital formulary or that insurance companies should cover the cost of a new drug in community pharmacies, requires a substantial investment by the company. For a hospital product, I have heard cost estimates from $8 million for a single-indication drug to $30 million for one with multiple approved clinical indications. For a community-based product, this cost could be much greater given the number of physicians, pharmacies and insurance plans one would have to inform.
A number of small companies have drugs in the pipeline in phase 2 or later development. They will all be severely challenged to meet the costs of launch if these drugs are actually approved for the marketplace. A substantial number could face bankruptcy just like Achaogen.
My proposal is that the various societies that promulgate authoritative clinical guidelines intervene either at the time of approval or simply on an annual basis to disseminate key educational information and very focused real-time clinical guidance as to how the new drug should be used. Every guideline has a table summarizing recommended therapies for the clinical indication under consideration. It is this table that could be targeted for modification. By focusing on just the therapeutic recommendations portion of the guideline, the task for societies should be less burdensome than a review of the entire guideline. These societies might include the IDSA, ATS, BSAC, the Society of Infectious Diseases Pharmacists and others. I am not sure how much this would affect initial expenses associated with launch, but it should accelerate uptake of deserving therapies.
To achieve this, sponsors would need to provide a full package of clinical trial, PK/PD and clinical microbiology data to the societies in a timely manner. I suggest that this would occur after the regulatory interactions that lead to approval. In this way, regulatory feedback can also be included in the information provided to the societies. Sponsors should also provide a preliminary price estimate to the societies. The societies would then modify (or not) the table of therapeutic recommendations in clinical guidelines for physicians. The societies would, of course, be free to provide additional details in guidelines that might expand on the table or even limit the use of the new drug based on their view of cost, side effects, toxicity and benefits compared to products already available. Of key importance would be to place the new product in the context of older therapies. (I have colistin and polymyxin firmly in my sights here). The sponsors would not pay the societies for this work. But, of course, sponsors would be free to disseminate any modified guidelines resulting from the societies’ work.
The societies, especially the ID pharmacists, should also consider making recommendations regarding off-label use. These should be based on clinical (if any), microbiological and pharmacodynamic and pharmacokinetic data.
 An example would be guidelines including a treatment recommendation table. This guideline should have been promulgated within a year after the approval of ceftazidime-avibactam. It might have discouraged the use of colistin or polymyxin for the treatment of resistant Gram-negative infections and encouraged the use of the newer, albeit more expensive, agent for cUTI, cIAI, and nosocomial pneumonia when resistant pathogens susceptible to ceftazidime-avibactam were suspected or documented.
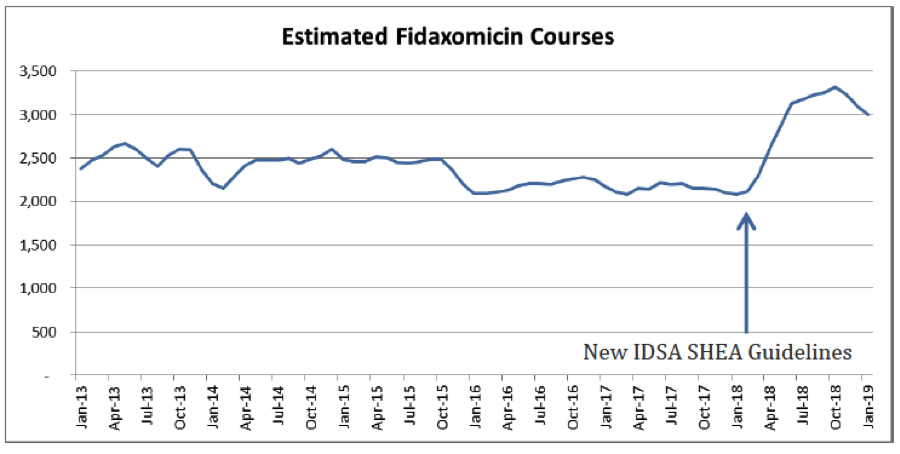
An example would be guidelines including a treatment recommendation table. This guideline should have been promulgated within a year after the approval of ceftazidime-avibactam. It might have discouraged the use of colistin or polymyxin for the treatment of resistant Gram-negative infections and encouraged the use of the newer, albeit more expensive, agent for cUTI, cIAI, and nosocomial pneumonia when resistant pathogens susceptible to ceftazidime-avibactam were suspected or documented. I think that we can all agree that timely modification of guidelines from experts would go a long way towards modifying the treatment paradigms utilized by physicians. If you have any doubt about this, look at the effect of a recent guideline change on the use of fidamoxcin in the treatment of C. difficile. Its shameful that it took eight years to get to this guideline change. Our expert societies simply have to do a better job of keeping up with the changing therapeutic environment as resistance evolves. Such action would, in a modest way, provide an additional pull incentive for sponsors and investors.
Category