
Contrary to populist dogma, with rare exceptions, chemicals do not accumulate in our bodies. For example, the plastic component BPA (1), a creation of Satan himself, according to certain academics and slimy environmental groups, has been used since the 1960s for a wide variety of purposes (2). So, the chemical is ubiquitous in the environment and can be detected in almost everyone's urine.
The key word here is urine. BPA and thousands of other chemicals and drugs end up in our urine because of our natural "detox" (3) system–the liver. Your liver is exquisitely designed to metabolize chemicals in such a way that they become more water-soluble (4) and pass through the kidneys on their way to the bladder where they are eliminated. This is why we aren't all a bunch of walking superfund sites.
HOW TO COLOR YOUR URINE, INTENTIONALLY OR OTHERWISE
I don't know whether you'd use this technology as a party trick but if so, it would work pretty well. Because if you slipped any of the three following items into the punch (5) you will probably scare the s### out of a bunch of people because the colors I'm talking about are not subtle.

Urine colors that will scare the hell out of you.
Here are the chemicals (two are drugs) that will make you pee in living color.
1. Methylene blue (green pee)
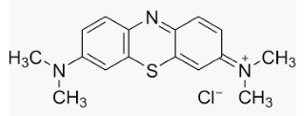
Methylene blue is one insane drug. Here are a few reasons:
- The name doesn't make any sense. In the parlance of organic chemistry, "methylene" universally refers to a CH2 group (methylene chloride is CH2Cl2). You will notice that there are precisely zero of them in the molecule. So, naming it "Rectal Itch Blue" or "Steve" would be just as accurate. Its real name is methylthioninium chloride.
- It is REALLY blue. How blue?

Methylene blue makes the guys in Blue Man Group look like albinos. The flask on the left probably contains far less than one gram dissolved in one liter of water. Although you can't see through it in this photo, since it is a solution, it is (by definition) clear (not colorless - these have different meanings), so with a strong enough light behind the flask (the sun?), you could see through the solution.
The serial dilutions above give you a better idea of its "blueness." At a concentration of 10 mg/100 mL (0.01%) the color is easily seen (1 mg = 3 grains of salt, 100 mL is about 3 ounces). But the blue can still be seen at a concentration that is 1000-times more dilute, an otherworldly low concentration of 0.00001%. To put this in perspective, the concentration of salt in the ocean is 3.5%. So, methylene blue is visible at a concentration that is 350,000-times less than salt in the sea. Photo: Longe City
- Methylene blue is considered to be the first synthetic drug, first used in the 1890s to treat malaria. It is still used today for methemoglobinemia - a dangerous condition, usually arising from certain toxins, where too much of a dysfunctional form of hemoglobin (methemoglobin) is formed. Methemoglobin doesn't carry oxygen properly. Methylene blue is also rather toxic, which is why it is no longer used for a dozen other conditions as it was in the past.
- Methylene blue is sometimes added to beer for St. Patrick's Day. The result? Bright green pee. Clever? Not so much.
2. Pyridium (Azo, phenazopyridine, orange pee)
Pyridium is a niche drug, but a very useful one. It has one use - as an anesthetic to relieve pain and burning during urination that affect people who have infections of inflammation in the urinary tract. These can make peeing a lot less fun than it ought to be.
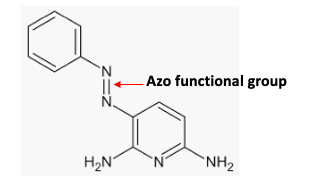
The chemical structure of pyridium. The nitrogen-nitrogen double bond is called an azo group and is responsible for the orange color, hence the brand name Azo.
Some of the oldest known organic chemicals are the azo dyes. This is because they are very easy to synthesize and, depending on the rest of the chemical structure, can produce a dazzling array of colors. They are used as artificial colors and for dying fabrics.
Pop a couple of Azo pills and the effect is not subtle. In about 20 minutes your urine will be seriously orange. It's difficult to describe how orange. Maybe radioactive Tang?
Pyridium is rapidly eliminated; 90% of it is gone within 24 hours. About half of the drug is eliminated unchanged and the rest if metabolized and then excreted in the urine.
While researching the pharmacokinetics of pyridium, I ran into a claim that was so bizarre that I was sure it was wrong:
Following oral administration, approximately 90% of the dose is eliminated in the urine within 24 hours, about 40% as unchanged drug & 50% as aniline & its metabolites, mainly p-aminophenol & N-acetyl-p-aminophenol (acetaminophen). (?!?!?)
No way, right?
In order for pyridium to be metabolized acetaminophen, it would have to go through a series of metabolic steps. that is so convoluted that Rube Goldberg would be envious. This had to be wrong. Except it's not. This is exactly what happens. (6,7).
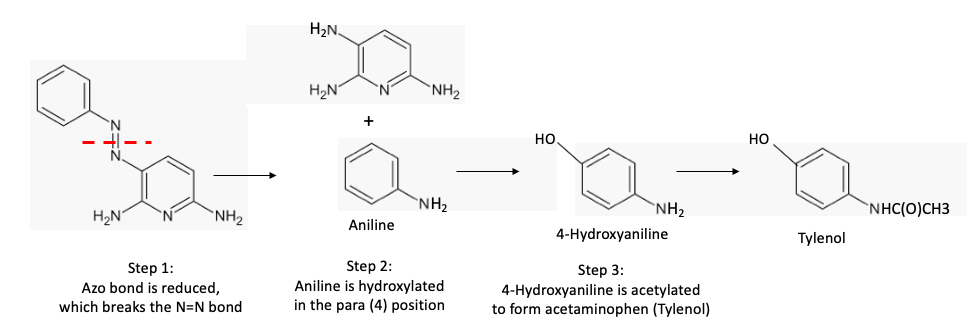
Metabolic madness. One of the metabolites of pyridium is another drug, Tylenol. Talk about convoluted.
3. Betanin (pink pee)
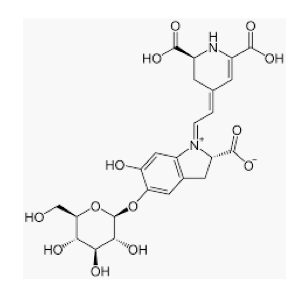
The chemical structure of betanin.
This happened to yours truly and it scared the crap out of me, at least, until the organic chemist mindset kicked in. Organic chemist or not, it is more than a little disconcerting to step up to the urinal and have red urine come splooting (8) out. Except the color didn't look quite right for blood. There was a pinkish hue to the red. Then I remembered that I had (for reasons I cannot explain) eaten a beet salad the night before. This had to be what was going on, it was just a matter of which chemical in the beets was responsible. A quick (very) trip to my office answered the question. The beet chemical that made rose-colored pee (a condition called beeturia) is called betanin, aka Beetroot Red.
The "lower" part of the betanin is a molecule of glucose. It can easily be removed, giving another red dye called betanidin, aka E162, which is a commonly used food additive.
Why are these things colored?
I've written about conjugation - consecutive alternating single and double bonds in a molecule - before (See: Color Yourself Busted: Organic Chemistry, Colors And 'Speed Traps'). Without going into detail, suffice it to say that when conjugation occurs so does color. It just so happens than all three pee colorants have five pairs of conjugated bonds. Here is a look at the three dyes. The alternating single (blue dot) and double (red dot) bonds are connected and consecutive. The number of conjugated bonds, as well as the presence of other atoms (especially nitrogen) determine the color of the dye. 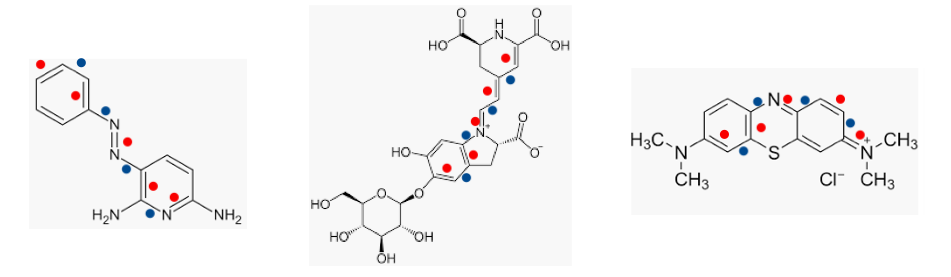
So, there you go. Pretty colors, a not-too-hideous chemistry lesson, party tricks, and maybe a trip to jail, all courtesy of your friendly neighborhood ACSH chemist. (And if you like this article feel free to send us another type of green. Just saying.)
NOTES:
(1) BPA is short for bisphenol-A, one of the two components used to make polycarbonate plastics. Despite protestations to the contrary, it is safe as used. See BPA Is Just As Dangerous As It Never Was.
(2) A common use for BPA polycarbonate plastics is food can liners. The plastic helps seal the can, preventing food poisoning.
(3) When you see a product or service for "detoxicification" it's a con. Eating some stupid fruit or squirting coffee up your anus to cleanse your body is quackery. Your liver cleanses your body. Let it do its job.
(4) To make chemicals/drugs more water-soluble the liver either breaks them into smaller pieces or adds solubilizing groups (sulfate, glucuronic acid...) to the molecule. The first example is called Phase I metabolism. The second is called Phase II.
(5) Don't. I'm not bailing you out.
(6) All three types of metabolism, azo reduction, para-hydroxylation, and acetylation, are known and fairly common. I just didn't expect to see the three of them in a row forming Tylenol.
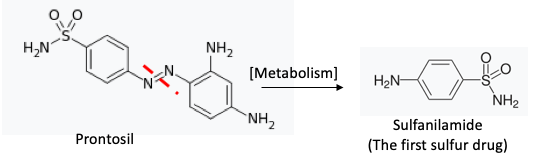
(7) The metabolic reduction of an azo dye resulted in the first non-arsenic-containing antibacterial drug. Although penicillin was discovered first (1928) Prontosil, a red dye, beat it to the market by seven years (1935) because of the difficulty of obtaining sufficient quantities of penicillin. Prontosil is a pro-drug of sulfanilamide - the actual antibacterial drug, which is a white solid. Sulfanilamide hit the market in 1936.
(8) Yes, splooting is a word. I know this because I made it up.



