Although different chemists may have other opinions, to me, the most important of all chemical bonds is an amide bond. It consists of a nitrogen atom connected to a carbon atom, which is doubly bound to oxygen (Figure 1).
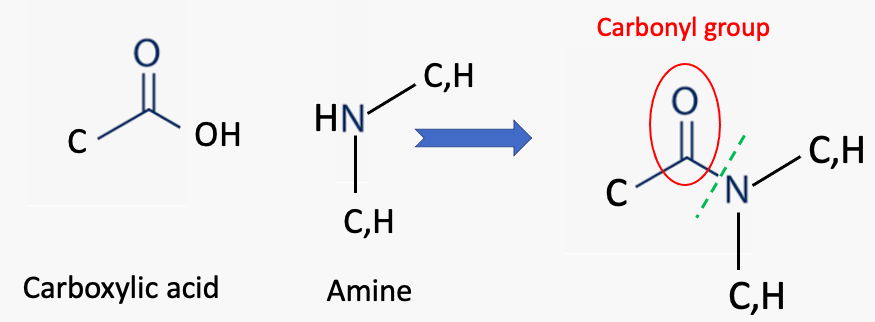
Figure 1. (Left) An amide group consists of a carbon atom (1) bonded to a carbonyl group (red oval), which is connected to a nitrogen atom. The nitrogen atom can be bonded to either carbon or hydrogen atoms.
Things look a little different, but the concept is the same for proteins; the difference is that the amide bond is formed between the amine of one amino acid and the carboxylic acid of another one (Figure 2).
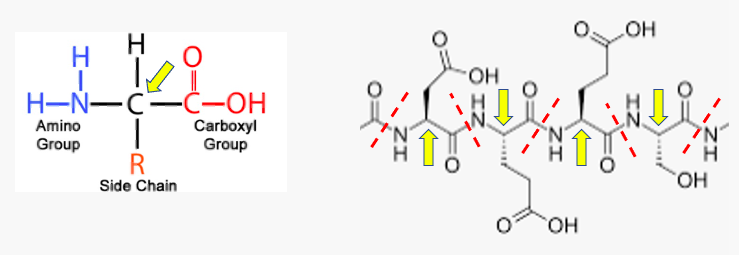
Figure 2. The basic structure of all amino acids that make up proteins. (Left) Amino acids, as the name implies, contain both amine and carboxylic acids in the same molecule. There are 20 different amino acids that make up proteins, all differing only by the side chains. Amino acids are so biologically important because contain both amine and carboxylic acid groups, enabling them to react with themselves to form polymers. These polymers are proteins – the basis of life. (Right) A small fragment of a protein containing 4 amino acids. The amide bonds are indicated by red hatch marks. The yellow arrows indicate the carbon where the side chains are attached. The largest protein in the human body contains 28,000 amino acids. Image (left): Astrochem
What does this have to do with COVID, Pfizer, or anything else?
This is where it gets interesting. When viruses replicate, they need to make copies of their genetic material and also must manufacture the proteins needed to provide structure and function for new virus particles. Thousands of amino acids are converted into large proteins by cellular ribosomes – protein factories. Although the ribosomes are perfectly happy to do this job (as if the dumb bastards have anything better to do), they can't finish the job.
When viral protein is made by the ribosome, it comes out as one long strand called a polyprotein. Polyproteins are useless to the virus. Although they contain all the structural and enzymatic proteins necessary to make new viruses, they must first be "processed" – cut into smaller pieces, which the virus can then use to replicate. This processing is done by enzymes called proteases. Pfizer's Paxlovid (2) inhibits this process, which is why the drug works; it is a direct-acting antiviral drug (DAAD) that is an inhibitor of the Mpro enzyme that cuts up the COVID polyprotein, allowing viral replication to take place.
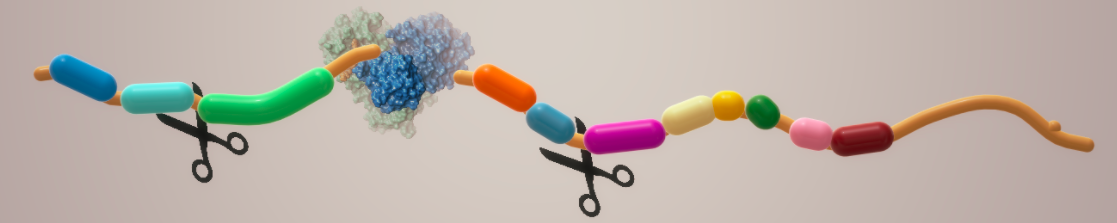
Depiction of a protease at work. The scissors represent the enzyme breaking amide bonds of a polyprotein, forming smaller (and useful) proteins. Image: Biova
One property of amides is that they're chemical tough guys; it is not easy to break the carbonyl-nitrogen bond. The reaction to break this bind is called hydrolysis, and you have to really pound on the molecule to make it happen. For example, conditions required to hydrolyze an amide include boiling it with strong acids (hydrochloric or sulfuric) or strong bases (sodium hydroxide) for hours to complete the reaction; protease enzymes can perform the same reaction millions of times faster. This is why drugs that inhibit these enzymes are so effective in shutting down polyprotein cleavage and, therefore, viral replication.
Pfizer's drug loves sulfur
There are four different types of protease enzymes. If I try to explain all of this, the two of you who are still reading this hideous article will cheerfully hurl yourselves in front of a bullet train. Since I don't want to lose such a large percentage of my followers, I'll keep it short and simple.
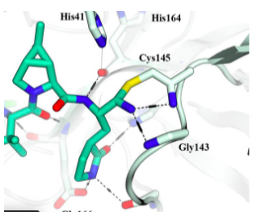
The COVID Mpro (main protease) is a cysteine protease, meaning that it uses the thiol (S-H) group on the amino acid cysteine to catalyze the cleavage of the protein's amide bond. Paxlovid is designed to inhibit this process by reacting with the carbon designated with a green star (below).
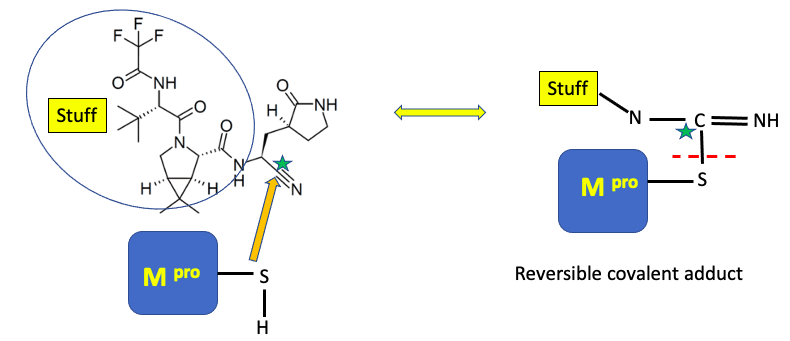
What's going on here is not as bad as it seems. The sulfur of the main protease reacts with (orange arrow) the nitrile (CN) group (green star) of the drug. Without going into too much chemistry, the nitrile is both chemically reactive and held within easy bonding distance of the cysteine sulfur atom (3). This results in the formation of what's called (sorry) a reversible covalent adduct (right), which means that a covalent bond, albeit a reversible bond, is formed between the inhibitor and the enzyme – a very good way to deactivate enzymes. A red hatch line designates this reversible bond. So, while Paxlovid doesn't permanently deactivate Mpro, it does a damn fine job of tying it up for a while.
I've sort of disrespected the rest of the molecule (blue oval) by calling it "stuff." But the stuff is also important; it causes Paxlovid to bind to Mpro and places the nitrile (cyano) group in the correct position to entice the enzyme's cysteine group into bonding with it. It wouldn't be inaccurate to call the "stuff" a "molecular wingman."
Bottom line
It is no accident that Paxlovid works; it is designed to do so. This is how drug design works: define an essential viral target (Mpro) and build a molecule to slow it down. This combination of chemistry and enzymology is both simple and complex at the same time and a fine example of how a drug can be rationally designed.
NOTE:
(1) There is one exception – hydrogen can replace carbon. These amides are called formamides.
(2) Paxlovid is actually a two-drug combination: PF-07321332, the actual inhibitor and ritonavir, originally an HIV protease inhibitor that inhibits the CYP enzymes that metabolize PF-07321332, giving it a longer half-life. For the purposes of this article I'm using Paxlovid and PF-07321332 interchangeably even though this is not technically correct.
(3) If you really want to torture yourself, here is the x-ray structure from a Pfizer paper which proves the mechanism of action. Good luck figuring this one out. And don't bother me!