There are at least two (1) scenarios in which COVID becomes "scary" (as in 2020 scary) again. We're already witnessing one at this time; the other is theoretical and has not yet come to pass.
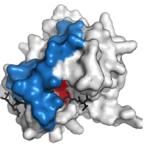
main protease
A recent, scary article in the Boston Globe proclaimed the following:
It's not totally absurd to compare our war against COVID with a boxing match. The virus clearly won Round 1; aside from masks and isolation, we were pretty much defenseless.
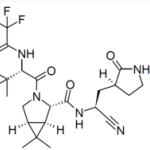
Although different chemists may have other opinions, to me, the most important of all chemical bonds is an amide bond. It consists of a nitrogen atom connected to a carbon atom, which is doubly bound to oxygen (Figure 1).
The "COVID era" is reteaching us a lesson that was learned in the 1990s and 2010s – that an effective direct-acting antiviral (DAA) medication can work wonders in controlling or eliminating a serious viral infection.
As Professor Katherine Seley-Radtke (1) and I wrote last July in an op