While many medications are simple to dose and sex-independent, many drugs are complicated, and very specific diseases require that doses be calculated based on mass, age, and many clinical parameters, including organ function. These established and validated algorithms include hundreds of different, complex formulas covering thousands of the over 20,000 FDA-approved drugs. They also necessitate a binary specification of male or female sex to calculate the correct answer.
The answer isn’t simply to have a "third" option for gender or to have algorithms for all 112 (and counting) socially or medically "manufactured genders." The only practical workaround requires biological sex on a patient's medical records.
Sex-Specific Medical and Drug Dosing Algorithms.
Pharmacokinetic (PK) formulas were developed to tailor medication dosages to an individual’s physiology. Although tailored to particular physiologic parameters (including sex) and even using well-researched PK/mathematical precision, it often still results in adverse reactions. According to the FDA, adverse drug reactions are already the fourth leading cause of death. Therefore, "requiring" the use of one's non-biological "preferred gender pronouns" in any clinical setting represents an additive risk of confusion to what is already an objectively sizeable source of morbidity and mortality.
"Preferred Genders" are Only Skin Deep. Sex-Based Physiology Endures Forever
There is no argument anywhere that a person's sex is determined at conception and that fertilization of either an "X" or "Y" sperm cell will instantly determine gender. Following conception, the divergent development pathways of males versus females are immediate, substantial, diverse, and enduring down to a cellular DNA level.
In humans, a single gene can contain approximately one million base pairs of DNA, and an "XY" chromosome (denoting male biological sex) or "XX" chromosome (representing female biological sex) can contain about 1,000 such genes. Every single cell in the human body (with a nucleus) has two sex-specific chromosomes, guiding their development and biological function. Indeed, sex is more than just "skin deep." It persists down to a microscopic, cellular level of the human body's 100 trillion (or so) cells.
It, therefore, doesn't matter how much pharmaceutical-grade synthetic estrogen or testosterone a person pumps into their body or how many medical/surgical procedures take place: the indelible imprint of biological sex -- determined in utero and at the moment of conception --is not alterable. A simple blood or tissue test at any point in one's lifetime (or even after death) will always reveal an XY or XX.
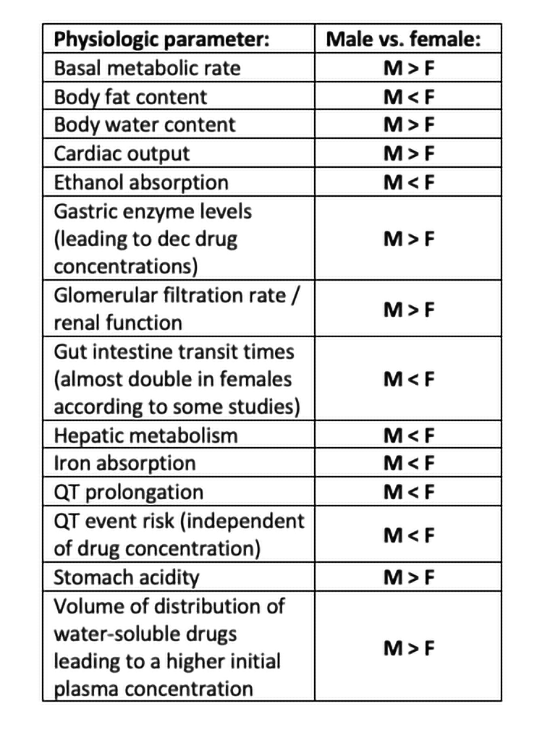
Published literature details an abundance of specific biologic, physiologic, and pharmacokinetic sex differences directly related to physiology, organ function, clinical pharmacy, drug development, basic science pharmacology, and the practice of medicine.
Some critical sex-specific parameters are summarized in the table below:
Estrogen's Effects on Drug Metabolism
It’s a common misconception that biological females simply need less of a drug than biological males because of their smaller mass. Estrogen levels in females are well-known to affect drug metabolism, especially on any medication utilizing the common CYP4503A, 2C19, or CYP2A6 hepatic pathways. (70-80% of all drugs in clinical use go through the CYP4503A paths alongside estrogen).
Estrogen and its additional effect on hepatic enzymes represent a significant additive problem to an already complex medical, pharmacokinetic, pharmacogenomic, and pharmacodynamic considerations. Additionally, "transgendering" individuals going through lengthy transitions require highly varied -- but still "superhuman" levels of estrogen -- which must be given to biological males to counteract biological testosterone levels. For instance, normal male testosterone levels are exceptionally broad and may vary anywhere from around 200 to 1,200ng/dL (i.e., a 500% difference).
Pharmacological and Physiological Complexity Require Biological Sex References
Today, around one hundred very commonly used sex-specific formulas can be found throughout medicine and pharmacology to calculate clinical parameters, risk, and correct drug dosages. These formulas cover most medical specialties and give one a sense of the variety and volume of formulas that require a binary specification of biological sex.
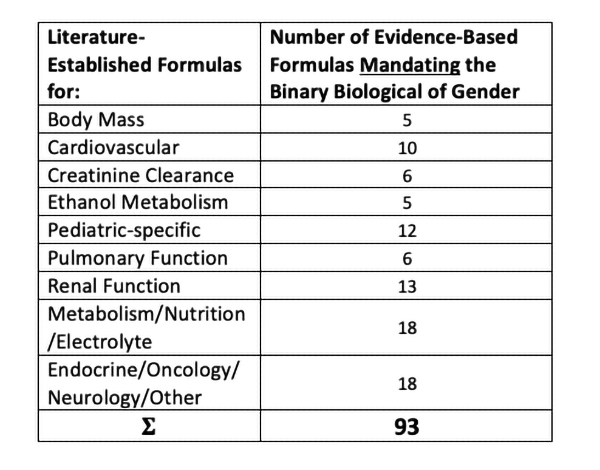
A complete, hyperlinked, and fully referenced list of sex-specific pharmacology and medical equations independent of this article may be viewed as a standalone addendum here.
Each existing medical, physiological, and pharmacology formula were developed following decades of carefully designed long-term research studies collectively involving hundreds of thousands of patients of all different genetic backgrounds to account for inter-individual variations.
Accommodating those who wish to defy the biological binary adds a massive level of complexity and would not just be as simple as adding a "third category" or stating "unknown." Because gender transitioning is such a highly varied stepwise undertaking, it would be impossible to re-work algorithms based on what chemical transitioning stage one is currently undergoing fighting their chromosomal biology.
On top of biological sex are the even more complicated metabolic differences between various races of people. There is an abundance of literature dating back decades detailing the wide genetic variations in the metabolism of estrogen- or testosterone-modulating drugs found in everyone -- even within phenotypically similar races such as Caucasian or Asian.
Creating models to generate new calculations to accommodate very high doses of estrogen or testosterone and generating new calculations for those who identify as transgender plus racial and inter-racial variability would have so many variables that it would become practically impossible.
The transgender population only makes up a small fraction of one percent of the people in the U.S., making it an expensive enterprise benefiting a few at a time when similar sums can be spent within our public health system to help many more.
In conclusion, biological gender must be mandated on all medical records to accurately determine body mass parameters, dietary intake, disease risk, drug dosing, and most measures of organ function.
[1] Frequently Asked Questions about Transgender People National Center for Transgender Equality
Sources:
Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation Pharmacology and Therapeutics PMID: 23333322
Gender Differences in Pharmacokinetics and Side Effects of Second Generation Antipsychotic Drugs Nervenarzt PMID: 16874502
Gender Differences in Pharmacokinetics of Alcohol Alcoholism Clinical and Experimental Research Alcohol Clinical and Experimental Research PMID: 11329488
Gender Differences in Pharmacokinetics. US Pharmacist
Gender differences in the effect of cardiovascular drugs: a position document of the Working Group on Pharmacology and Drug Therapy of the ESC European Heart Journal PMID: 25948737
Gender Effects in Pharmacokinetics and Pharmacodynamics Drugs PMID: 8521756
Gender-related differences in pharmacokinetics and their clinical significance Journal of Clinical Pharmacology and Therapeutics PMID: 10583696
How Important Are Gender Differences in Pharmacokinetics? Clinical Pharmacokinetics PMID: 12036391
Pharmacogenomics, pharmacokinetics, and pharmacodynamics: interaction with biological differences between men and women British Journal of Pharmacology PMID: 23981051
Sex- and Gender-Based Pharmacological Response to Drugs Pharmacological Review PMID: 33653873
Sex-related differences in pharmacokinetics and pharmacodynamics of anti-hypertensive drugs Nature PMID: 22089536




