More than 60 years after its discovery, the cancer drug vincristine still works. It just doesn't work in the same way that has been taught to physicians and cancer researchers for decades. The "old" mechanism of action by which a class of antiproliferative drugs was thought to function is similar but also distinctly different from that which a group from the University of Wisconsin-Madison elucidated and recently published.
Although to the average reader, this discovery may seem trivial, perhaps even unimportant, in the cancer world, this is more like an apple flying off Sir Isaac Newton's head and ending up back on the tree. That's because there is a critically important class of chemo drugs called microtubule poisons – something I coincidentally (1) discussed in my last article. (See: If You're A *Poison Expert* It Is Unwise To Poison Your Wife. A Colchicine Lesson.)
Unsurprisingly, ChatGPT hasn't "caught up," but otherwise did OK.
Microtubule poisons disrupt the normal dynamics of microtubules, either by stabilizing them to prevent depolymerization or by destabilizing them to prevent proper polymerization. These disruptions interfere with critical cellular processes, particularly cell division.
Inhibiting mitosis
Microtubule poisons can be divided into two classes of drugs: Those that promote microtubule stability (microtubule-stabilizing agents) and those that do the opposite (microtubule-destabilizing agents). Both classes inhibit cell division – one of the foundations of cancer therapy.
Microtubule-stabilizing agents include the commonly used taxanes, (paclitaxel (Taxol) and docetaxel (Taxotere).
Microtubule-destabilizing agents, also commonly used, include the vinca alkaloids (vinblastine, vincristine, vinorelbine, and others) and colchicine, which is less commonly used because of its toxicity. People taking vincristine are "lucky" in the sense that it is in the minimal emetogenicity category, the lowest category for the incidence of vomiting after a single IV dose, less than 10%. By contrast, cisplatin, which I wrote about recently, is in the highest risk category (>90%).
A mind-blowing experience
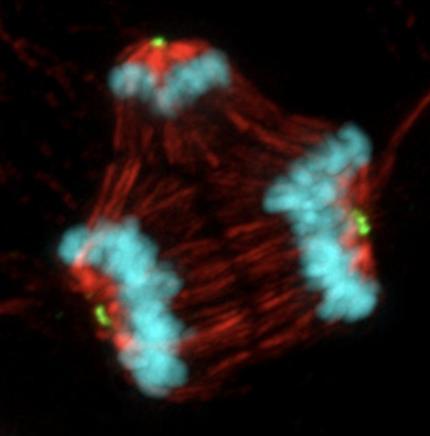
The Wisconsin scientists studied tumor samples taken from breast cancer patients at the University of Wisconsin Carbone Cancer Center. All patients had been treated with typical mitotic poison chemo drugs like vincristine or Taxol. The group measured how much of the drugs made it into the tumors and observed how the tumor cells responded to different concentrations. They found that some cells continued to divide after exposure to the drug but did so abnormally, the Eureka moment. At certain drug concentrations, the mitosis process "went nuts." Multiple clusters of daughter cells, having as many as five new cells, all sharing one set of chromosomes, were found (Figure 1). This was the group's mind-blowing experience.
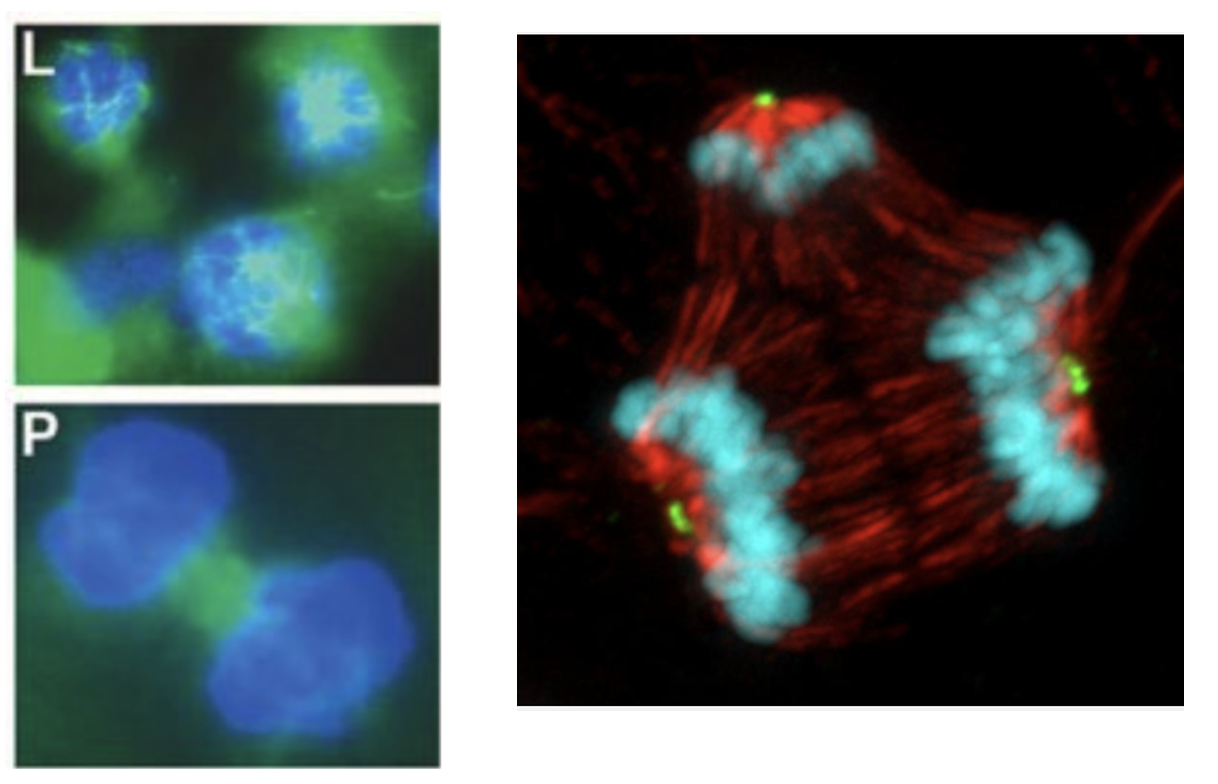
Figure 1. (Left) The "old" theory on microtubule mechanism of action. The figure shows stained cells treated with vinblastine (top) and untreated (bottom). (Right) A cell treated with paclitaxel divides into three genetically different daughter cells, which may or may not be viable. Credit: Beth Weaver, University of Wisconsin–Madison, PLOS Biology
“This was sort of mind-blowing...For decades, we all thought that the way paclitaxel works in patient tumors is by arresting them in mitosis. This is what I was taught as a graduate student. We all ‘knew’ this. In cells in a dish, labs all over the world have shown this. The problem was we were all using it at concentrations higher than those that actually get into the tumor.” [emphasis mine]
Dr. Beth Weaver in SciTechDaily
Time to rewrite textbooks?
It would appear that there are two problems with the long-held tenet regarding the mechanism of action of mitotic poisons: 1) Mitotic poisons don't necessarily stop cell division; they can also "mess it up," killing some cancer cells (this is why the drugs work) but allowing others to continue dividing in strange and previously unseen ways. 2) The effect seems to be a function of the concentration of the drug within the cells, which is lower than previously believed.
“So, after mitosis you have daughter cells that are no longer genetically identical and have lost chromosomes... We calculated that if a cell loses at least 20% of its DNA content, it is very likely going to die."
Dr. Beth Weaver, ibid
Now what?
This knowledge could substantially reshape cancer research. Should cancer scientists continue to look for superior antiproliferative drugs, or are they on the wrong path? If there is an inherent flaw in the understanding of how mitosis disrupters work, perhaps it's time to reconsider. Dr. Weaver says yes:
"We've been barking up the wrong tree...We need to refocus our efforts on screwing up mitosis — on making chromosomal segregation worse."
(1) "Coincidence" doesn't even begin to describe how strange the timing is here. My Ph.D. involved the synthesis of vincristine analogs (microtubule poisons) as potential cancer drugs. About 50,000 years ago.