
Just another day at the office.
1. Go to Long Island off the coast of the Bahamas. Coordinates (23°41.12′ N, 75°22.18′ W) are included, in case you happen to be still using Apple Maps, because you are just as likely to end up here:
Apple Maps: Still not all that reliable
2. Get into one of these things.

A Johnson-Sea-Link I manned submersible. Not for claustrophobes.
3. Submerge 630 meters (2067 feet, seven football fields).
4. Look for an unknown sponge. This is not as easy as you'd think when you're 2067 feet below the water in something that looks like a shop vac with a glandular condition. Which sponge would you pick?
Photo: Biological Education
5. Get the stuff out of the sponge:
The sponge specimen was frozen at −20 °C immediately after collection and stored frozen until extracted. The frozen material as described above (255 g) was extracted exhaustively with ethanol by macerating in a Waring blender, filtering off tissue and re-extracting the tissue a total of 5 times (2.5 L total). The extract was concentrated to a dark orange oil (15.9 g) by distillation under reduced pressure.
This may not sound so bad, but this does:
A total of 46.5 mg was obtained. Extrapolating back to wet weight of sponge, Dragmacidin G is a major component of the sponge and is present at 0.36% of wet weight of sponge.
If that strikes you like a lot of work to get 1/123rd of the weight of a teaspoon of salt from 16 pounds of an unknown sponge, you are right. But it may be well worth it. Dragmacidin G, the name given to the active chemical in the sponge is both very unusual looking (structurally) and also has some intriguing antimicrobial properties.
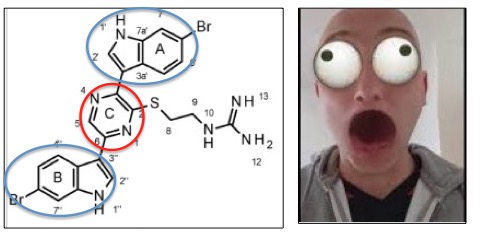
Dragmacidin G— a chemist's nightmare.
Dragmacidin G has a unique chemical structure—two 6-bromoindole groups (blue circles), each connected to a pyrimidine ring (red circle). While the image on the left may not have much meaning to you, any organic chemist will tell you that trying to solve (determine) the chemical structure of the abomination of the left will turn you into the guy on the right.
The amount of work this alone required, even with the most modern analytical instruments is your basic nightmare. Just to give you an idea...
Structure Elucidation

NMR analysis used to establish the structure of Dragmacidin G. Don't try this at home.
(Note: Since organic chemists use these same basic techniques to solve structures of run-of-the-mill compounds, we are at least somewhat proficient in doing so. But not with this bad boy. This one is like kicking an 80-yard field goal into the wind, wearing ballet slippers.)
Was it worth the effort? It would seem so. Finding new classes of antibiotics is as important an endeavor as any in pharmaceutical research.
(See: "A New Class Of Antibiotics From Lichens? Maybe.")
The article above is a few weeks old, but is useful for contrasting and comparing the two discoveries. The following table shows the antibacterial activity of the lichen antibiotics: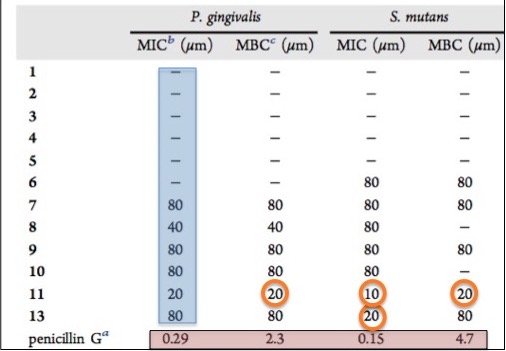
The antibacterial activity of compounds isolated from lichens. MIC values are expressed in micrograms/mL
I noted in the earlier article that while lichens did contain two chemical compounds that had antibacterial properties, they were not potent enough to become drugs. The orange circles indicate the MIC (minimum inhibitory contraction—the concentration of drug required to stop bacteria from growing—of compounds 11 and 13 against two different oral bacteria. By comparison, the reference sample penicillin G had MIC values ranging from 5-10 times lower, meaning that penicillin G is 5-10 times more potent than the lichen-derived compounds. For antibiotics with MICs in the 20-80 microgram/mL range to be effective, the required dose would have to be huge. Probably enough so to be toxic.
But, the table below tells a radically different story.
The MIC values of six well-known antibiotics from five different classes are given. Here you can see resistance with your own eyes by comparing the concentration of the antibiotic required to kill both susceptible and resistant versions of Staphylococcus aureus and Methicillin-resistant Staphylococcus aureus—the infamous and deadly MRSA*.
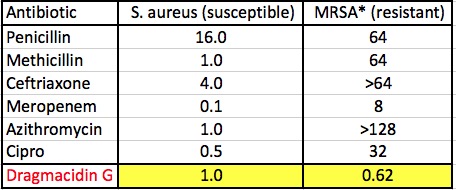
Antibacterial activity of common antibiotics (in micrograms/mL)
Source: http://www.antimicrobe.org/b237tabrev.htm
Penicillin, which is rarely used, and methicillin belong to the penicillin class. Ceftriaxone is cephalosporin (1). Meropenem is a carbapenem (2). Azithromycin (Zithromax) is a macrolide, and Cipro is a fluoroquinolone. These are critically important antibiotics that were previously used to wipe out staph infections, but cannot do so anymore, as indicated by the 50-100 fold loss of activity against MRSA. Now look at Dramacidin G. It kills not only staph but MRSA too—something that other the six drugs can no longer do.
Will Dramacidin G make it to the pharmacy at your hospital? There is a long way to go. Toxicity is one of many potential pitfalls. But the discovery of a new class of antibiotics that can kill MRSA is quite an achievement by any measure.
Notes:
(1) Ceftriaxone is the last line of defense against gonorrhea. (See: The coming gonorrhea epidemic, New York Post, September 5, 2012)
(2) The even scarier infection, carbapenem-resistant enterococcus (CRE), which has a 50 percent mortality rate is resistant to Meropenem and other carbapenem antibiotics.
Original Source: Wright, A Mar. Drugs 2017, 15(1),



