If you buy a frozen leg of lamb at the supermarket how do you know whether it has thawed at some point from the provider to the market and then refrozen? Currently, there is no way to know, so consumers have no choice but to assume that the meat has been kept frozen the entire time. But, thanks to some clever chemistry from a group at the Ministry of Agriculture & Farmers Welfare in Matsyapuri, India, there may be a simple way to monitor whether the food has been subjected to "temperature abuse," which could cause spoilage. The method requires a tiny amount of gold and chitin, a readily available and biodegradable polysaccharide (1) which is found in crab and shrimp shells. The chemistry is straightforward and the results can be seen with the naked eye.
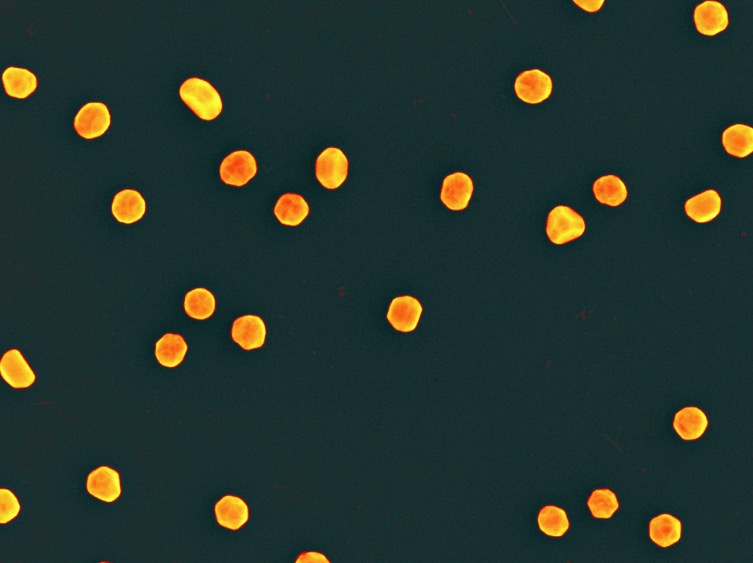
Gold Nanoparticles. Image: Popular Science
Chitradurga Mohan and colleagues recently published the results of a pilot study in Science of Food (A Nature journal) in which the group converted a gold solution (1) into gold nanoparticles (AuNP), which serve as an indicator of temperature abuse (2). Here's how it works.
- Chitin, the second most abundant natural polymer (cellulose is first) is too insoluble to react with gold(III) chloride trihydrate (HAuCl4•(H2O)3), the aqueous gold solution, but the group found that chitosan, a close chemical relative of chitin does the job very well. Chitin can be converted to chitosan by a simple deacetylation reaction (Figure 1).
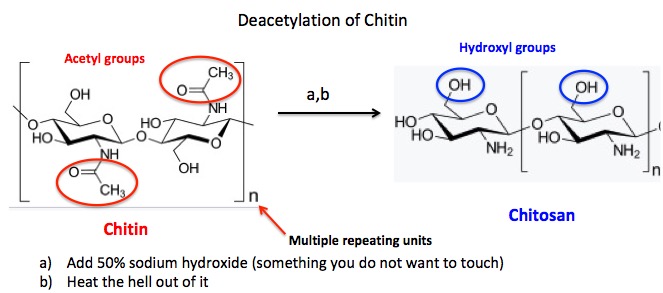
Figure 1. Deacetylation of chitin
2. Chitosan reacts with Au III to give Au0 aka elemental gold (Figure 2)
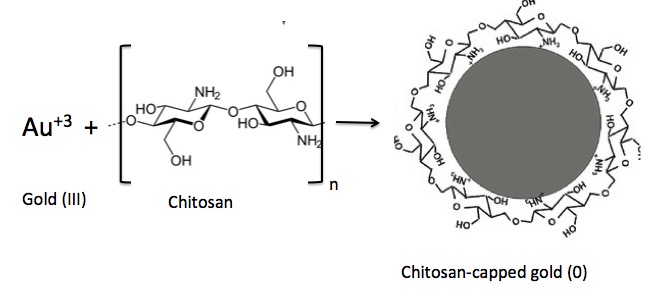
Figure 2. Reduction of gold (III) with chitosan to give metallic gold metal (Au0) "capped" by chitosan molecules via interaction with positively charged protonated amino groups.
3. How it works.
It is difficult to see In Figure 2, but the chitosan cap is bound to the gold nanoparticle by positively charged protonated amino (NH3+) groups. Here is a small portion of the diagram enlarged. The red arrow indicates the NH3+ group that binds to the gold.
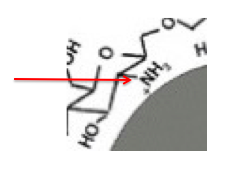
These positively charged fragments repel other positively charged particles; this helps prevent the particles from aggregating (forming a cluster). But at different temperatures aggregation can still take place and this can be seen with a color change. When first formed, the particles give the solution a red color. But with time, aggregation takes place and the color changes from red to blue to purple. The darker the color, the more aggregation.
The authors believe that the chitosan-capped gold nanoparticles could be placed in packaging act as tiny biosensors- thermal history indicators. To simulate this they mixed the gold (III) with 0.25% chitosan in glass wells - it turns bright red - and froze the solutions. The frozen material was subjected to an "abused temperature) of 37ºC (98.6ºF) and observed the color change over 24 hours. After a few hours, the color change becomes evident.
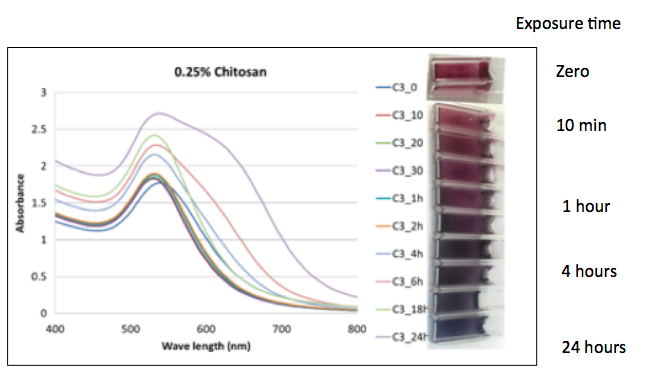
Figure 3. Effect of temperature abuse on the color of chitosan-capped gold nanoparticles at different time intervals (10 min to 24 h). Source: Science of Food.
The authors conclude:
Chitosan, a biopolymer of crustacean waste can be used as a reducing agent for the synthesis of gold nanoparticles, which is a greener method. Variation in the concentration of chitosan results in varying the size of AuNPs and exposure to temperature- abused condition resulted in a distinctly different color indicating its efficiency to use as [thermal history indicators] . This can be used to ensure the quality and safety of frozen stored perishable food and pharma products during shipment and transportation to distant locations. The preparation method is very simple and ecofriendly and easily adoptable by the industry.
A very interesting method of using nanotechnology and chemistry to address a public health issue.
NOTES:
(1) Since gold is inert (unreactive) it is not common to see gold salts, such as gold (III) chloride. But gold does react with aqua regia - a mixture of nitric and hydrochloric acids. The gold "dissolves" forming gold (III) chloride. It goes without saying that aqua regia is not something you want to mess with.
(2) Gold nanoparticles have been made by this method
Original paper: C. Mohan, S. Gunasekaran, and C. N. Ravishankar "Chitosan-capped gold nanoparticles for indicating temperature abuse in frozen stored products" npj Science of Food V3 (2019). DOI: https://doi.org/10.1038/s41538-019-0034-z




