According to the World Health Organization, there are vaccines available to prevent 26 infectious diseases. Notably absent on that list is norovirus - the so-called "stomach flu." Given that norovirus is one of the most contagious of all viruses, causing 20 million cases per year in the US - second only to the common cold - a vaccine would be of substantial benefit to public health.
Although deaths in the US are uncommon - less than 1,000 per year - worldwide the "stomach bug" is responsible for 200 million cases and about 50,000 deaths in children every year. So, it is not surprising that vaccines against norovirus have been the focus of considerable research (for example, here and here). The CDC has published a review on the global burden of norovirus and the prospects for vaccine development.
But a vaccine against norovirus has been a tough nut to crack (1); the factors involved with protection against infection are not well understood, but they do involve human blood type, something I wrote about this past winter (See Do You Get The Stomach Bug While Others Dodge It? It Could Be Your Blood). Furthermore, the virus mutates readily; there are more than 25 different strains of norovirus. Multiple strains and rapid mutation are both barriers to vaccine formation.
Most of the mutation of the virus involves the highly variable P2 (P is short for protruding) domain on the surface of the viral capsid (Figure 1).
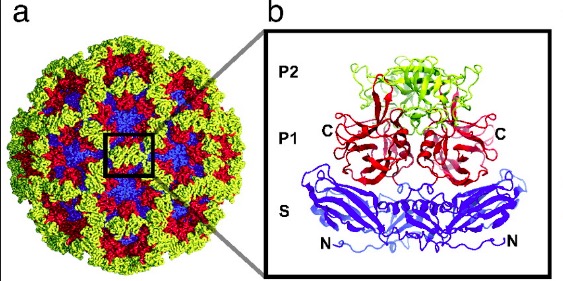
Figure 1. The P2 domain on the surface of the norovirus capsid is "hypervariable" (readily mutates), which leads to structural changes in the capsid that cause it to no longer bind to the antibody that makes up the vaccine. "P" is short from protrusion, meaning that this exposed part of the viral capsid is responsible for binding the virus to its target receptors. This explains why the mutation of the P2 part of the capsid can defeat neutralizing antibodies. Image: PNAS
One strategy to inhibit pathogens that readily mutate is to target a portion of the virus that stays constant. This is called a "conserved region." The concept is not new, but now a group of researchers from the University of North Carolina at Chapel Hill, the University of Texas at Austin, and the NIH Vaccine Research Center has applied it in efforts to find a norovirus vaccine.
Three adult volunteers were injected with an experimental vaccine. Blood samples were collected and an antibody called A1431 was identified. A1431 inhibited the binding all of the known strains (dating back to 1987) of norovirus-GII.4 (2) - to the receptors of target cells (3). GII.4 has been the predominant family of circulating viruses for decades and is responsible for 60% of all outbreaks and also the most serious infections.
The reason that A1431 inhibited all GII.4 strains was that it bound to a conserved region of the viral spike (4) in all GII.4 strains of norovirus. This is the most intriguing finding in the paper; it suggests that the process of viral mutation, which can render an existing vaccine ineffective, can be overcome by targeting a region of the virus that does not mutate. An antibody that binds to a conserved region will offer stronger and longer protection, thus, a better vaccine.
"For a vaccine to be efficacious against pandemic GII.4 strains, it must be able to train the immune system to focus on the part of the GII.4 virus that does not change over time."
Ralph S. Baric, PhD, School of Global Public Health, University of North Carolina
NOTES:
(1) By contrast, there are two effective vaccines against rotavirus, which is sort of a kid's version of norovirus.
(2) Norovirus can be divided into genogroups - related viruses within the genus. There are five norovirus genogroups, GI-GV. GI, GII, and GV infect humans. GII.4 is short for Norovirus Genogroup.4 Sydney - a strain of the virus first discovered in Australia in 2012. It is the cause of most norovirus outbreaks.
(3) Norovirus targets epithelial cells in the small intestine.
(4) Viral spikes are protein structures that are found on the outer coating of the virus. They are responsible for the binding of the virus to its host cell
(3) The mapping information was obtained by analyzing the antibodies that were formed by injecting three adult volunteers with a vaccine candidate.