One of the many fascinating aspects of organic chemistry is how seemingly different reactions work in the same exact way. There can be no better example of this than phenomenon than two very different functions – saving victims of Tylenol poisoning and neutralizing the smell of skunk spray – operating by an identical mechanism - a chemical reaction called a Michael addition.
Robert H. Cichewicz and colleagues at the University of Oklahoma have been looking for natural products that could "de-skunk" people and pets and they have a candidate called pericosine A - a metabolite that was isolated from sea hare-derived fungus Periconia byssoides.
Pericosine A
If you're asking "what the hell does this have to do with Tylenol poisoning, I must regretfully inform you that you must endure the...

Tylenol itself is harmless, but it is converted by the liver to a toxic metabolite called N-acetyl-p-benzoquinone imine, aka, NAPQI. When too much NAPQI builds up in the liver it does irreversible damage to the cells. It is a bit ironic that Tylenol would be much safer if our bodies did not process it in a way to make it more harmful than the drug itself.
So, what do Tylenol and pericosine A have to do with each other? If you look a little closer, similarities start to appear (Figure 1).
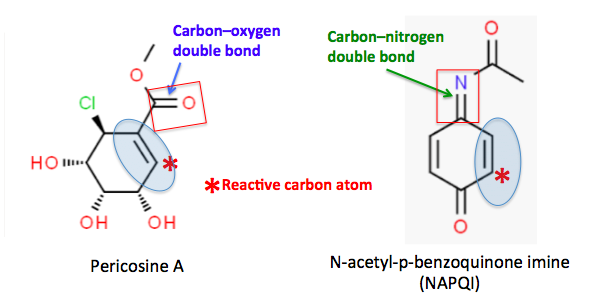
Figure 1. (Left) Pericosine A contains a carbon-carbon double bond (blue oval) connected by a single bond to a carbon-oxygen double bond (also called a carbonyl group, red rectangle). For reasons you can't possibly want to know, this makes the carbon atom marked with the red asterisk chemically reactive, especially toward thiol (carbon bound to a sulfur atom, which is bound to a hydrogen atom) groups. (Right) NAPQI also contains a carbon-carbon double bond (blue oval) connected by a single bond to a carbon-nitrogen double bond (red rectangle). As is the case with pericosine A, this makes the analogous carbon atom (red asterisk) chemically reactive, again, especially with thiols.
So, it is not surprising that both pericosine A and NAPQI will react with ubiquitous sulfur-containing compounds called thiols. Oddly, in one case the thiol is the antidote, while in the other case, the thiol is the "culprit." This can also be explained by organic chemistry.
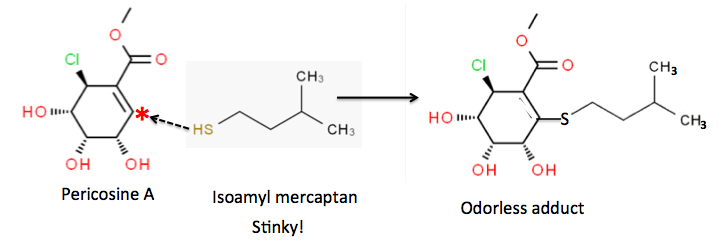
In the case of skunk odor, it is pericosine A that is the "antidote." This is because compounds of this type of compound (1) reacts with the vile-smelling low molecular weight thiols that skunks use as a defensive mechanism. One of the primary skunk thiols, isoamyl mercaptan, reacts with pericosine A to form an odorless stable compound (called an adduct). The reaction between sulfur and carbon is denoted by the black hatch arrow.
In the case of Tylenol poisoning, things are reversed; the thiol (called N-acetylcysteine) is the antidote.
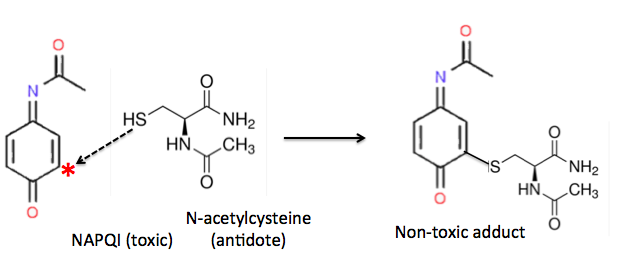
Ain't chemistry grand? A compound from a marine fungus may end up being an anti-skunk spray and a chemical like the skunk spray prevents people from dying from a Tylenol overdose. And both reactions work by the same mechanism. The things you learn.
According to the authors the pericosine A seems to be non-irritating to the skin, so it's possible that we may have a product that will de-stink your kid or pet by using a Michael addition – a 140-year old reaction. (2)
NOTE:
(1) When carbon-carbon double bonds are attached to carbon-oxygen double bonds they are called alpha, beta-unsaturated carbonyls. When the second fragment is a carbon-nitrogen double bond they are called alpha, beta-unsaturated imines. In both cases, the molecules are reactive at the carbon atom furthest from the oxygen or nitrogen atom (red asterisk).
(2) The Michael addition was invented in 1887 by Arthur Michael at Tufts University.
----------------------------------------------------
Original reference: R. Cichewicz, et. al., J. Nat. Prod. Publication Date: July 5, 2019 DOI: https://doi.org/10.1021/acs.jnatprod.9b00415




