
Never let it be said that ACSH isn't at the forefront of reporting critical topics. No siree, if something important breaks, we're on it. And fast.
It is not terribly surprising that human behavior would change in the midst of an unprecedented global health threat. This has resulted in a variety of curious behaviors, some sane and others, not so much. For example, it's not unreasonable to have a full freezer. After all, panic creates more panic, which can lead to food hoarding. So if you're worried about running out of an essential staple like pimentos (1) by all means, fill up the basement with cartons of the damn things.
My household is (marginally) saner than that of the pimento hoarders. We decided to stock up on a bunch of useful stuff in case something like COVID-20 makes an unexpected appearance. One of these items was milk. Now, if you're wondering why a 172-year-old man needs a bunch of milk, all I can say is that Dr. Pepper – vile in its own right – and Honey Bunches of Oats do not make a savory combination. So I needed a bunch of milk. Some of it went into the refrigerator and the rest into the freezer.
Recently a container was removed from the freezer and thawed, and the excitement began! You'd think as a chemist and milk aficionado that I might have been prepared for what came next. Wrong!
The stuff that came out of the carton looked like spit-up. It did not taste like spit-up, although I can't say for sure since it's been a while since I've tried any. The milk was weird looking but smelled and tasted fine. Easy solution. Shake it up. The result? Something that looked like shaken spit-up. The evidence is below.

Left: Honey Bunches of Oats bathed in spit-up milk. Center: Close up. The red circles indicate areas of excessive spit-up-age. Right: Spit-up milk poured into the sink in case the other subtle images don't fully capture the magnitude of the event.
What is going on?
One explanation might be that the milk spoiled, but this isn't what happened. It smelled like milk, not sour milk, which is formed by a chemical reaction. Chemical reactions are quite sensitive to temperature; cold slows them down. A rule of thumb is that a reaction rate will double when the temperature rises by 10oC. Working backward, the temperature in a refrigerator is about 4oC, while a freezer is about -18oC. So, all things equal (2), milk in a freezer should last about 4-times longer than in a fridge.
If the change wasn't chemical, it had to be physical. It was. I had unknowingly managed to reverse the homogenization of milk, a process discovered almost 100 years ago.
Fat and Protein Don't Mix
Just like oil and water, fat (non-polar) and protein (polar) don't mix. This is why milk is icky until it is homogenized. During the process, the globs of fat that don't mix with milk (mostly water and protein) are turned into tiny particles. Homogenized milk is an emulsion – a mixture of two components that don't dissolve together. Here's one way that homogenization is done industrially.
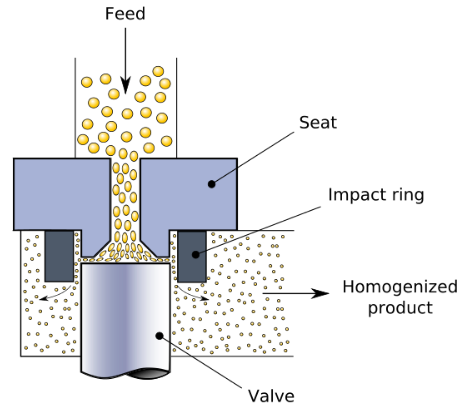
High-pressure homogenization of milk. Source: Wikipedia.
Here's the process at the molecular level.
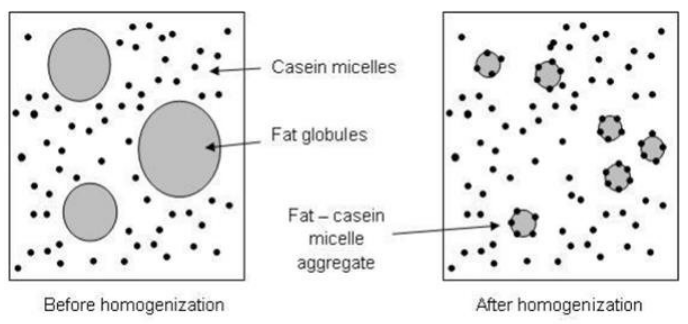
(Left) Fat in milk "seeks" other fat and forms globules, which separate from the protein and water in the milk. (Right) Homogenization breaks the globules into tiny particles which are coated with casein (milk protein). Source: Biosystems and Food Engineering Research Review 21, May 2016, Edition: 21, Publisher: University College Dublin, Editor: Enda J Cummins, Thomas P. Curran, ISBN: 1649-475X
What the Hell Happened to the Milk?
Freezing and thawing cause milk to separate, essentially becoming "un-homogenized." The fat separates into small globules, which give the milk its spit-up-like texture. There's nothing wrong with it, but the grainy texture completely skeeved me out, especially the ickies floating around in my coffee.
Almost, but not quite enough to make me spit up.
NOTES:
(1) Just my opinion, but pimentos are pure evil.
(2) All things aren't really equal. A low temperature will slow a chemical reaction, but a change in its physical state can make even a bigger difference. Solids are much more stable than liquids or solutions. This is why most pills are still OK well past their expiration dates.



