I'm always skeptical of claims concerning therapeutic uses of CBD oil (cannabidiol) and also the results of clinical trials that contain a small number of participants. So when I came across a 2023 paper in the Journal of Dental Research (peer-reviewed) about a clinical trial with 61 participants that examined whether CBD could be used to treat toothache pain, I was tempted to ignore it. But the trial by the Rutgers School of Dental Medicine should not be ignored; it was a double-blinded placebo-controlled randomized clinical trial (RCT) with two different doses of the drug – the so-called "gold standard."
The Rutgers groups divided 61 patients who were suffering from moderate to severe toothache into three blinded randomized groups:
- Placebo
- Low dose CBD-treated (10 mg/kilograms body weight (mpk))
- High dose CBD-treated (20 mpk) (1)
The primary outcome was the numerical reduction in pain score, while there were several secondary outcomes, including the amount of time until relief and the force with which participants could bite. The CBD used was Epidiolex (2), the brand name for a solution of pharmaceutical CBD, which was approved by the FDA for certain types of epilepsy in 2018.
Summary of results
Pain
Figure 1 (below) shows the magnitude of pain reduction measured by the Visual Analogue Scale. Although there seems to be a considerable difference in pain in the CBD-treated participants, note that statistical significance is only seen at 120 and 180 minutes in the high-dose group and 180 minutes in the low-dose group. Although the authors claim that the onset of relief is 15 minutes for the high-dose group and 30 minutes for the low-dose group, neither the graph nor the statistics seem to support this claim. It must be noted that pain in the placebo group also declined, so the magnitude of the effect in the treated groups must be adjusted downward by the placebo group.
Conclusion: If any real pain reduction occurs, these data neither prove nor disprove it.
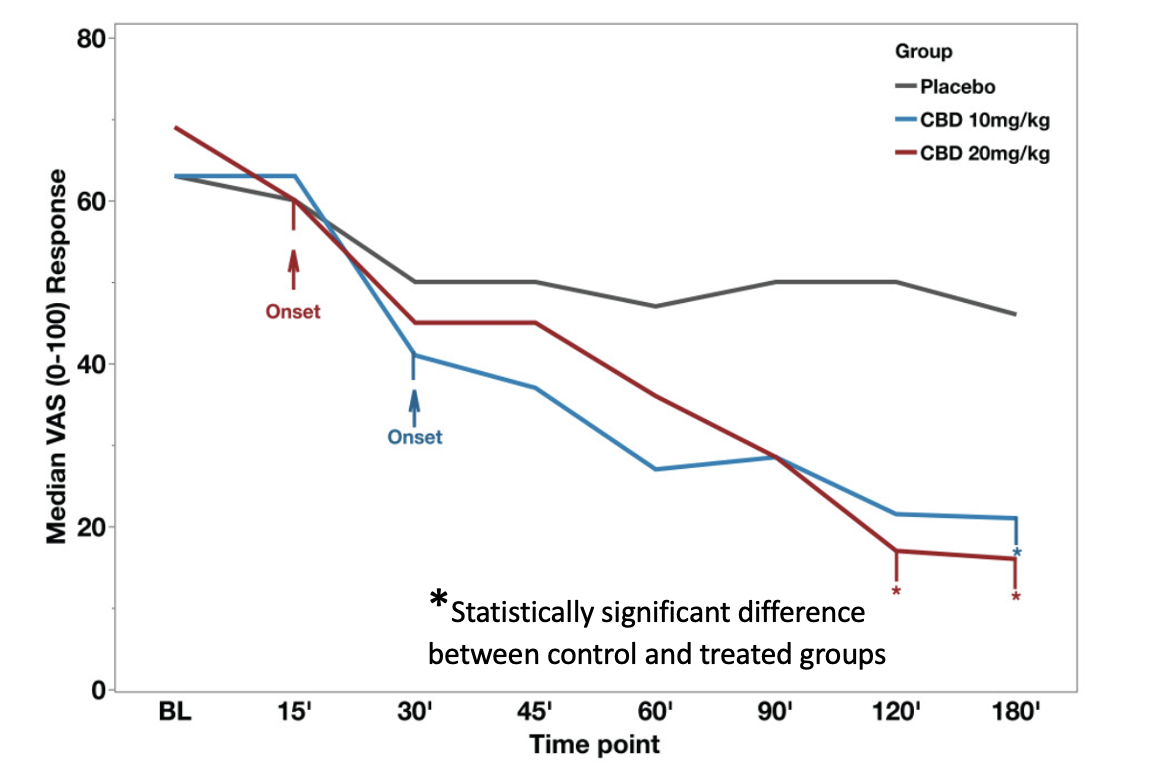
Figure 1. Median Visual Analog scale pain scores at different time points. The red and blue arrows indicate the onset of pain score relief from baseline (BL) for the cannabidiol (CBD) groups. Asterisks designate statistical significance. There are only three such data points, all very late in the measured time interval. Source: Chrepa, et. al., "Cannabidiol as an Alternative Analgesic for Acute Dental Pain." Journal of Dental Research, Online first 11/1/23. https://doi.org/10.1177/0022034523120081
Force of bite
Although biting on something with an atomic tooth may not sound as pleasant as, say, a day spa treatment, it is nonetheless one of the measures used to determine the potential reduction of dental pain by a drug. These data are shown in Figure 2.
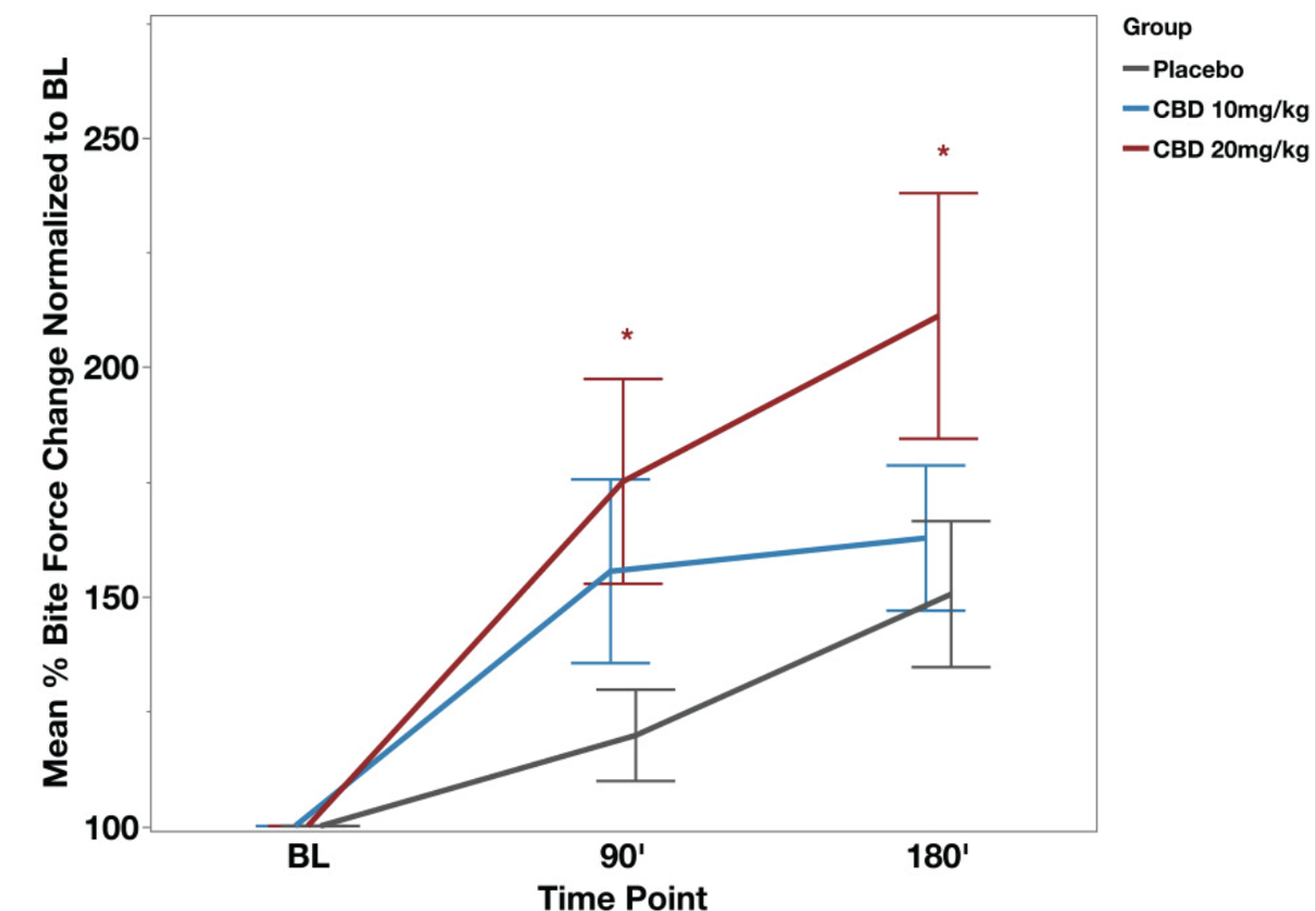
Figure 2. Bite force as a function of two CBD doses.
At 90 and 180 minutes there is statistical significance between the high-dose and placebo groups. However, there is no significant difference between the low-dose and placebo groups at any point in time, so there is no way to demonstrate dose dependence, a strong indicator of a pharmacological response.
Conclusion: This graph is more convincing than that in Figure 1. The effect of high-dose CBD appears to be real. The low-dose treated group may or may not be experiencing relief, but keep in mind that this group (10 mpk) is receiving half the FDA-recommended dose for patients with the two types of epilepsy for which it is approved.
Bottom line
All in all, CBD may have a useful effect, but the evidence in this study is far from robust. Nor is it evident that the drug fails entirely. It may be that a higher dose of CBD will prove to be effective, although an increase in side effects is probably inevitable (3). A larger study, which is probably worth doing, will be necessary to tighten up the statistics and possibly define a protocol with a sufficient difference between placebo and drug-treated patients and determine which if any, dose can provide pain relief with acceptable adverse effects.
NOTES:
(1) For epilepsy, the FDA-recommended dose is 20 mpk.
(2) Epidiolex is the brand name for a CDB solution made by Greenwich Biosciences Inc., Carlsbad, CA
(3) The drug has numerous side effects, especially gastrointestinal. These effects (not shown) were: 20 mpk >10mpk > placebo. It is not clear whether pain patients could tolerate a significantly higher dose. (20 mpk equals 1.4 grams per day in a 70 kg person.)




