
Dr. Wallace, thank you so much for agreeing to answer some questions that arose from my article Pain Relief And No Ulcers? 'Magic Aleve' In Clinical Trials about Antibe's ATB-346.
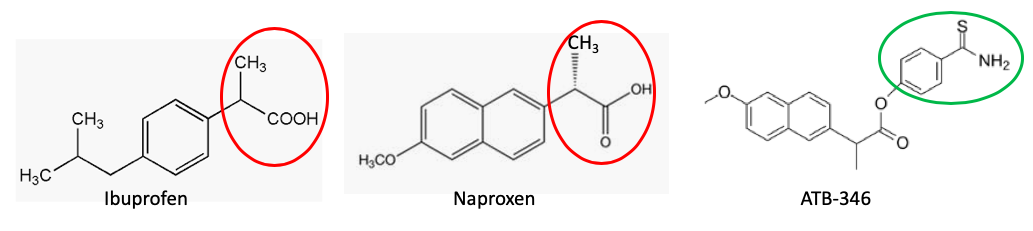
JB: Naproxen and ibuprofen are chemically identical at the “business end” (red circle) of the molecule. The thiobenzamide group (green circle) that gives ATB-346 its unique properties would presumably work the same with ibuprofen as the parent drug. Why did you select naproxen instead of ibuprofen?
JW: Naproxen is regarded as the most cardiovascular-safe of the NSAIDs (this was first shown when the selective COX-2 inhibitors were introduced, and serious CV adverse events were being observed)
JB: My readers and I both wondered about the short interval for developing ulcers. I’ve always been under the impression that gastric ulcers take years, not weeks, to develop.
JW: Acute lesions are formed within an hour of taking an NSAID, but they are not considered clinically significant. True ulcers (which penetrate through a layer in the stomach/intestine called the muscularis mucosae can form in days of NSAID use.
JB: Do you know where in the body the second step – cleavage of the ester bond to release naproxen – takes place? Liver? Serum?
JW: We are not sure when the ester bond is cleaved. There is metabolism at the NSAID end of the molecule before the ester bond is broken, and H2S is released soon after absorption of the drug
JB: 250 mg of ATB-346 seems to work better than 1000 mg of naproxen. Do you attribute this solely to the action of the hydrogen sulfide?
JW: You should not think of the drug as two separate components. ATB-346 suppresses cyclooxygenase activity and releases H2S. It is ~6-times more potent, and once-daily administration produces beneficial effects for 24 hours. When we make an identical molecule to ATB-346 but without the sulfur, it is no longer “GI-safe” (ulcers form). Also, the dose (250 mg) is one-sixth of the daily naproxen dose. IT IS A DIFFERENT DRUG THAN NAPROXEN!
JB: Naproxen works for acute pain. Given the superior efficacy of ATB-346 after 10 days is there any reason why it wouldn’t also work for acute pain?
JW: We are confident that it will work for acute pain.
JB: Can you tell us anything about your early-stage ABT-352 and why it is being developed for acute pain? Is 352 based on the same platform as 346?
JW: ATB-352 is a hydrogen sulfide-releasing derivative of ketoprofen, a very powerful, but VERY GI-toxic NSAID. Our studies show that ATB-352 does not produce GI damage and is a very potent pain-killer. Part of the mechanism for the enhanced analgesic effects of ATB-352 is a significant contribution of endocannabinoids. I have attached a recently accepted publication about this drug.
JB: Although you have apparently “eliminated” the GI toxicity of naproxen, would you expect ATB-346 to have less kidney toxicity than naproxen? How about other NSAID side effects, such as allergies and tinnitus.
JW: Difficult to say. A once-daily drug will likely have safety benefits over twice-daily. We have not seen any adverse effects on blood pressure in extensive animal studies and the human studies to date at doses up to 750 mg – 3x our recommended dose. Allergies and tinnitus – I don’t know.
JB: Thanks, again. My readers and I will be keeping our fingers crossed that you and your team can pull this off. It would be quite an accomplishment.
#Dr. John Wallace is a Canadian pharmacologist with a long-standing interest in drug development for GI disorders. He trained in the UK with Nobel laureate Sir John Vane. He earned his Ph.D. at the University of Toronto and also has an MBA from the UK. He is the founder of Antibe Therapeutics Inc. and also serves as the Chief Scientific Officer.



