Convalescent Plasma
For those of us, unfortunate enough to be infected by COVID-19 but fortunate enough to survive the experience, our blood holds antibodies to the virus. Specifically, those antibodies can be found in serum – the liquid, non-cellular component of the blood. Using a technique called plasmapheresis, donors can have their blood siphoned off, centrifuged to remove the plasma along with the antibodies, and get their red blood cells back.
Perhaps you have seen the ads on television asking for plasma donations from individuals convalescing, recovering from COVID-19. They are the human source of convalescent plasma. It was approved for use in those seriously ill with COVID-19, the thought being that the additional antibodies would enhance a patient’s immune defense and improve outcomes.
A study by a consortium of academic medical centers led by the Mayo Clinic published the initial results of the use of convalescent plasma in the New England Journal of Medicine. These results led to the FDA’s restriction in its use.
The Mayo Study
The study consisted of adults, over age 18, with at least one risk factor for progression to severe COVID-19. [1] The outcome of interest, survival. The work involved 600+ hospitals across the country, ultimately recruiting 3082 patients. The patients were given convalescent plasma with various amounts (titers) of antibodies broken down into low, medium, and high levels. There were no control or placebo patients. The treated participants were 61% men, 23% Black, 37% Hispanic – all the usual risk we have come to expect. They were younger than we might anticipate; 69% were younger than 70.
- 29.6% of the low titer group died
- 27.4% of the medium titer group died
- 22.3% of the high titer group died
- Those individuals not receiving mechanical ventilation did better in each titer level than those on ventilators.
These mortalities are high but remember these are really among the most gravely ill with COVID-19. High titer infusions of convalescent plasma do reduce the mortality for patients not already on ventilators by 25% - it helped, but was a long way from an effective treatment for most of these patients.
There were some nuances. Patients treated earlier did better than those treated later; the difference was just a day. In the real world, it is difficult to predict who will take a turn for the worse, and giving everyone convalescent plasma is not possible given the limited supply. You might even argue that the report tries to find some glimmer of hope when little existed. Here is the plot showing the reductions in mortality in the high vs. low titer groups over time, with and without mechanical intervention.
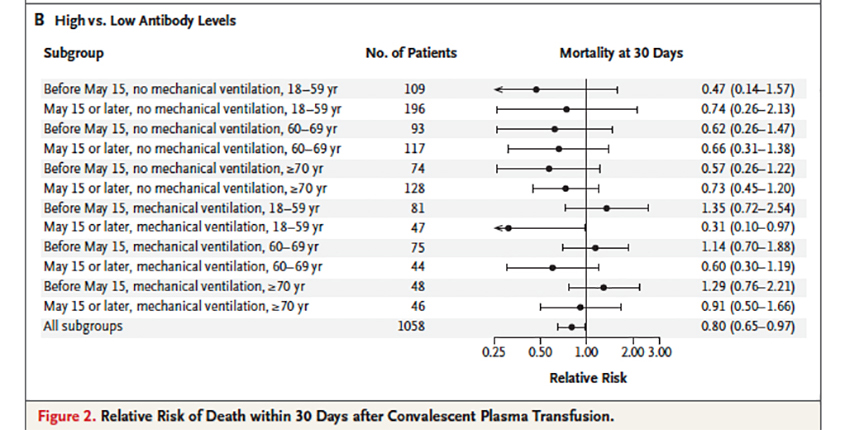
As you can see, the confidence intervals all cross the relative risk of 1, meaning no benefit or harm. While over 94,000 patients received these infusions, the data is based only on a third of the population.
“These data show that the benefit of convalescent plasma was most apparent in patients who received plasma transfusions containing higher levels of anti–SARS-CoV-2 IgG antibodies early in the disease course.”
Based upon these findings, the FDA restricted the use of convalescent plasma to high titers and patients early in their course, before requiring intubation and mechanical ventilation.
This was a good moment; science, available for all to read and consider, was used in making a regulatory decision.
[1] These risk factors include respiratory failure, dyspnea, desaturation of the blood, an abnormal respiratory rate, a large difference in oxygen indicating that oxygen is having difficulty going from the lung into the bloodstream, low overall oxygenation, or changes in lung X-rays –measures of respiratory problems. Additionally, patients would qualify if they had failing or failed heart or kidney function or septic shock.
Source: Convalescent Plasma Antibody Levels and the Risk of Death from Covid-19 NEJM DOI: 10.1056/NEJMoa2031893