
Following the FDA's granting of Emergency Use Authorization to Merck's molnupiravir and Pfizer's Paxlovid, the only two approved direct-acting antiviral drugs; we now have two badly needed tools to deal with COVID, especially since it is clearly not finished dealing with us. Although both drugs reduce hospitalizations and deaths from COVID, they each have plusses and minuses. Let's compare the two.
I. Ease of Synthesis
Below are the chemical structures of Merck's molnupiravir (left) and Pfizer's nirmatrelvir (right). (Nirmatrelvir is now the generic name for what used to be called PF-07321332, the antiviral drug component in Paxlovid. Paxlovid also contains a second drug called ritonavir, which prolongs the half-life of nirmatrelvir in the blood.)

Does anyone want to guess which of the two is easier to synthesize?
Given that it is unlikely that most of you know anything about organic synthesis, and even if you do, you're probably unemployed, your only hint is that Paxlovid looks a lot bigger and more complicated than molnupiravir, so intuitively, it's going to be harder to put together, Although this "method" of evaluating synthetic difficulty is, at best, naive, in this case, it happens to be correct. The original molnupiravir synthesis (there are now patents for several different versions ) consisted of five simple steps starting with uridine, a chemical that can be bought by the ton. It is very easy to make.
What about nirmatrelvir?
I don't really know, nor can I find anyone who does. One chemist friend, who is partly in the loop but wishes to remain anonymous, calls the synthesis "a bitch." Since I used to do this kind of thing professionally, all I can tell you is while I would cheerfully undertake the molnupiravir synthesis, I would probably hide under my bed if tasked with nirmatrelvir. Even if there were spiders under there. But Pfizer is a very large company with plenty of resources. They will be able to handle it.
II. Mechanism of Action
Both drugs inhibit viral replication, but in very different ways. Molnupiravir interferes with the assembly of new viral RNA, without which the virus cannot replicate. In this way, it operates a bit like remdesivir or other nucleoside inhibitors, but there's a big difference. For example, the herpes drug acyclovir works by tricking the viral DNA polymerase (the enzyme that puts together the DNA nucleobases required to build new herpes DNA) into incorporating it rather than deoxyguanosine, the nucleobase that would normally go in a specified position. Once acyclovir is incorporated, the polymerization process stops by a process called chain termination.
Molnupiravir acts a bit like acyclovir, but with a big difference. N-Hydroxycytidine (NHC), the active drug (molnupiravir is a pro-drug), tricks the RNA polymerase enzyme into incorporating the drug in place of either uridine or cytidine in a similar manner to how acyclovir "sneaks into" the herpes viral DNA. But it doesn't stop there.
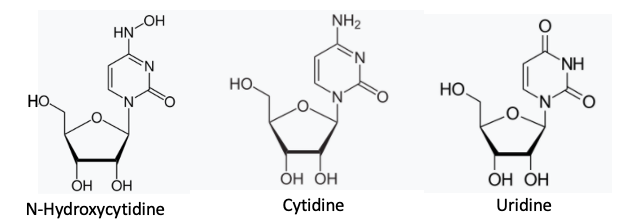
Instead, once NHC becomes incorporated, it does something insidious: More nucleobases (A, G, C, and U) keep adding to the growing RNA chain for a while, but in a crazy, random way – something that could be called "RNA on LSD." The incorporation process continues for a while, but when enough "incorrect" nucleobases are incorporated, the new RNA molecules are so mutated that the virus eventually undergoes what is called an "error catastrophe," leaving behind a bunch of non-functional mutant viruses. This raises a safety concern: Will NHC will also incorporate into human DNA and be genotoxic or maybe even carcinogenic? More on this below.
By contrast, nirmatrelvir inhibits COVID replication by inhibiting a critical viral enzyme called a protease, a method that has already been responsible for significant advances against both HIV/AIDS and hepatitis C, something I wrote about here. There are no molnupiravir-like safety concerns associated with protease inhibitors.
III. Efficacy
My celebration of molnupiravir's 50% reduction in hospitalization, which was based on interim data, was premature. Once the final data were analyzed, that number plummeted to 30%. This is not good. Not one bit.
Fierce Biotech's Nick Paul Taylor summed things up nicely:
The efficacy of Merck’s COVID-19 antiviral has fallen sharply. While most Americans were sleeping off Thanksgiving, Merck revealed the relative risk reduction fell from 50% to 30% in the final analysis of its phase 3 data, sending its stock sliding down...The drug looks less effective in the final analysis. After factoring in patients who became evaluable after the interim analysis, the rates of hospitalization or death in the molnupiravir and placebo arms were 6.8% and 9.7%, respectively, reducing the relative risk reduction to 30%.
Nick Paul Taylor, Fierce Biotech, November 29, 2021
And the FDA didn't come across as all that delighted in its EUA letter to Merck. Here are the criteria for its use:
- [People with] positive results of direct SARS-CoV-2 viral testing, and...
- Who are at high-risk5 for progression to severe COVID, including hospitalization or death, and...
- For whom alternative COVID-19 treatment options authorized by FDA are not accessible or clinically appropriate. (Hmm. Does this mean remdesivir, which is about as useful as a Wheat Thin, or sotrovimab, the one monoclonal antibody that works against omicron but is nearly impossible to get? No thanks, I'll take the Paxlovid)
By contrast, Paxlovid's interim data matched the final data:
Final data available from all high-risk patients enrolled in EPIC-HR study (n= 2,246) confirmed prior results of interim analysis showing PAXLOVID™ (nirmatrelvir [PF-07321332] tablets and ritonavir tablets) reduced risk of hospitalization or death by 89% (within three days of symptom onset) and 88% (within five days of symptom onset) compared to placebo; no deaths compared to placebo in non-hospitalized, high-risk adults with COVID-19...0.7% of patients who received PAXLOVID were hospitalized through Day 28 following randomization (5/697 hospitalized with no deaths), compared to 6.5% of patients who received placebo and were hospitalized or died (44/682 hospitalized with 9 subsequent deaths).
Pfizer press release, 12/14/21
In terms of efficacy, Paxlovid beats molnupiravir, and not by a little. So, why not just add molnupiravir to the waste stream and simply prescribe Paxlovid? The answer is: In drug research, it's never that simple.
IV. Baggage
No matter how efficacious or safe, every drug has something wrong with it. The same holds true with both molnupiravir and Paxlovid, but they have a different brand of baggage.
Molnupiravir
As I mentioned above, the knock on molnupiravir is that it is a mutagen. This does not mean that it's a carcinogen (especially for a five-day course - how many people get lung cancer from smoking for five days?) or will cause any harm at all. But mutagenicity must be taken seriously. Here are some highlights from Merck's 11/30/21 presentation to the FDA:
Molnupiravir is mutagenic in vitro (outside the body)
According to Merck, "Molnupiravir was mutagenic in vitro." This was based on three standard in vitro assays of mutagenicity (1).
But not so in animals
The gold standard of in vivo mutagenicity is the transgenic rodent assay. NHC, the active drug, was a clear negative in this test. In two other in vivo tests, the drug was negative in one case in inconclusive in the other. This led Merck to conclude "Molnupiravir has only a low risk of genotoxicity (DNA damage) in vivo."
A word of caution
Although in vitro (cell) mutagenicity does not necessarily result in genotoxicity (DNA damage) in animals or humans there is a caveat. Merck's assessment of genotoxicity is that it is "low risk," But the risk is higher, perhaps unacceptably so, in developing fetuses. Exposing developing fetuses to mutagens is a seriously bad idea. The warning is precautionary rather than data-driven, but it's hard to argue with the logic.
Hence, a 12/31/21 "Dear Healthcare Provider" letter issued by the FDA warns of fetal harm because of the possibility of mutagenicity [emphasis added]:
Molnupiravir is not recommend-ed for use during pregnancy. Based on findings from animal reproduction studies, molnupiravir may cause fetal harm when administered to pregnant individuals. There are no available human data on the use of molnupiravir in pregnant individuals to evaluate the risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes
The FDA also recommends that breastfeeding be discontinued during the molnupiravir treatment period for the same reason.
Paxlovid
Paxlovid comes with a different warning, although it has been overblown in the press. One of its two components, ritonavir is a powerful inhibitor of a liver enzyme called CYP3A, which metabolizes and clears drugs from the blood. Ritonavir's job is to keep nirmatrelvir in the blood longer. It does just this but will do the same for dozens of other medications. So the use of ritonavir can result in the elevation of the blood levels of other drugs people might be taking, possibly to dangerous levels. Some drugs, like statins or sedatives, can probably be discontinued during the treatment (or their dose adjusted), but with other drugs, for example, blood thinners, antiarrhythmics, and blood pressure medications, this may be risky. Your physician and pharmacist ought to be able to work out temporary dose changes or discontinuations during the Paxlovid treatment period.
V. What's the Better Drug?
The answer is that there is no right answer. While most people (including yours truly) will go for the 89% reduction in hospitalization (Paxlovid) vs. 30% for molnupiravir, people who take certain drugs may be unable to safely take Paxlovid (even though it has a very good safely profile) and opt for molnupiravir instead. But for pregnant women, molnupiravir is out of the question, and Paxlovid is the clear choice. As an asthmatic who is at fairly high risk, I will feel much better once I have a bottle of Paxlovid, not molnupiravir, in my pocket. There's a different risk-benefit equation for different people – just like there is with other drugs.
VI. Bottom Line
Having two antiviral drugs to choose from is a good problem, considering that we had none a month ago. Given that both drugs have issues, albeit different issues, the very large difference in efficacy between Paxlovid and molnupiravir is probably going to drive most patients and their physicians to Paxlovid. It's unfortunate that molnupiravir was not more effective. Having two highly efficacious drugs –both working by different mechanisms – to choose from would make life during these difficult times much easier. Nonetheless, these drugs will put a real dent in COVID and possibly turn into a nuisance rather than the horror show we see every night on the news.
Notes:
(1) In case you're a masochist, molnupiravir was mutagenic in both the Ames Test and Chinese hamster ovary assay but negative in the human micronucleus test. I do not get paid enough to try to explain this stuff.
#Disclosure of financial conflict of interest: My IRA contains Merck and Pfizer stock.



