Molnupiravir, trade name Lagevrio, is one of two COVID oral antivirals on the market, the other being Paxlovid. Molnupiravir is a pro-drug of N-hydroxycytidine – an oxidized analog of cytidine, one of the four bases that constitute RNA. Molnupiravir is converted into molnupiravir triphosphate (MTP), which is similar but not the same as cytidine. MTP is incorporated into the RNA of the virus during its replication, creating “errors in sequence fidelity,” for our purposes, just simple mutations that result in many of the viral replicants being dead on arrival. The clinical result is a dramatic reduction in viral load demonstrated in the laboratory, animal models, and us.
Molnupiravir has always played second fiddle to Paxlovid; it was just not as effective. [1] While its use was initially widely adopted, it has been supplanted by other therapies, and its use has declined. One early concern before its approval has been molnupiravir’s mutagenicity, especially for pregnant women. The newest concern is again about mutagenicity, but in this case, its effect on the virus. Simply put, “whatever doesn’t kill you may make you stronger.” The you, in this instance, is the virus.
A moment of science
The nucleotide analog, MTP, once incorporated into COVID’s RNA creates mutations, specifically, guanine to adenine, G-to-A mutations, and, to a lesser degree, cytosine to uracil, C-to-T mutations [2]. The presence of a large number of G-to-A mutations in the COVID genome is a “signature” of molnupiravir’s action.
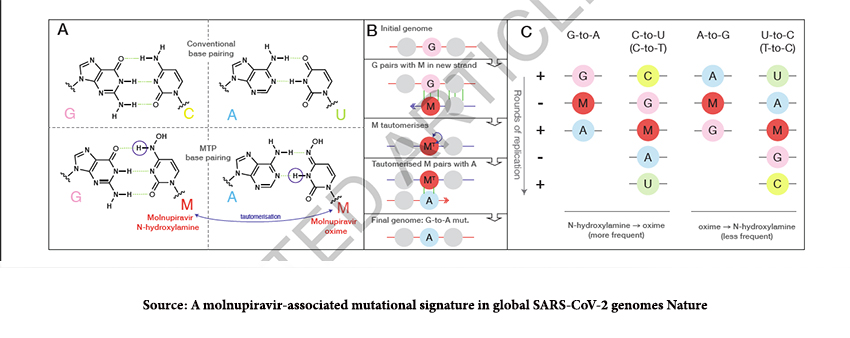 Researchers established molnupiravir’s signature by comparing COVID genomic data of untreated patients and those receiving molnupiravir to the “typical SARS-CoV-2 mutational spectrum.” As to be expected, molnupiravir,
Researchers established molnupiravir’s signature by comparing COVID genomic data of untreated patients and those receiving molnupiravir to the “typical SARS-CoV-2 mutational spectrum.” As to be expected, molnupiravir,
“led to an 8-fold increase … in the rate of mutations and that this increase was highly specific to transition mutations… especially to G-to-A.”
The researchers further visualized the area within the COVID genome using what is referred to as a “mutation annotated tree.” Let your hearts be still; it simply is a genome map, just like an ancestry tree you might purchase from Ancestry.com, where the branches are differing mutations rather than aunts and uncles. They identified a branch they felt represented the effects of molnupiravir on the viral genome. [3]
The signature they identified is both the strength and weakness of the study. It does allow the researchers to make some conclusions, as we will see in a moment, but as they write,
“Identifying a particular branch as possessing a molnupiravir-like signature is a probabilistic rather than absolute judgement.”
Moreover, their results pertain to “long” branches. For smaller branches, it was challenging to determine the cause of mutation, as chronic infections are also known to cause mutations.
Smoking Gun?
Molnupiravir’s signature branches:
- Were almost all sampled after the roll-out of molnupiravir in late 2021 and early 2022
- In many countries that used molnupiravir, the researchers observed mutations with a high proportion of G-to-A branches. In contrast, countries with a low number of G-to-A branches, like Canada and France, had not authorized the use of molnupiravir.
- In Australia, where molnupiravir was provided to nursing facilities and recommended for residents over age 70 testing positive for COVID, high G-to-A branches primarily occur in an aged population.
“These correlations between spectra from high G-to-A branches and known molnupiravir-treated individuals strongly support a shared mutational driver, and therefore that the high G-to-A branches are driven by molnupiravir treatment.”
A two-edged sword
I return where I began, “Whatever doesn’t kill you may make you stronger.” There is little doubt that using molnupiravir results in lethal mutations in the replicated virus, clinically resulting in lower viral loads that, unfortunately, are not as clinically helpful in reducing hospitalization and deaths. It might be useful in reducing transmission, but that is speculation for another day. But the mode of action, creating mutations, increases the mutational pressure on the virus and may result in finding a new “fitness” in the landscape – translated, a virus that could become more infectious, as we have already seen. While we have luckily seen a decline in the severity of illness, there is no guarantee that other mutations will not result in heightened severity.
On a purely statistical basis, without consideration of the biological environment, more mutations allow for greater exploration and increase the possibility of a more fit virus.
The study is just another example of the dance between COVID and us. We co-evolve with all within our environment. There is no evidence that molnupiravir has caused a mutation harmful to us, but we should be more humble in the presence of the technologies we have created and how they impact not just our intended target but all of us.
[1] A recent study published in JAMA Open Network found that Paxlovid seems to have waning efficacy, but not to the extent of molnupiravir.
[2] I am unsure why scientists refer to uracil as T rather than U, but to be true to the article, that is their phrasing.
[3] For those who absolutely must know, these branches had “at least 10 substitutions, of which more than 25% were G-to-A, more than 20% were C-to-T and more than 90% were transitions.”
Source: A molnupiravir-associated mutational signature in global SARS-CoV-2 genomes Nature DOI: 10.1038/s41586-023-06649-6




