Given the drama around the last two drugs approved for Alzheimer’s dementia, Aduhelm and Leqembi, we have a right to be skeptical.
The first one approved, Aduhlem, demonstrated little, if any, clinical effect. Its “accelerated” approval in June 2021 came over the objection of the FDA advisory group, prompting several resignations and severe criticism. Subsequently, irregular interaction between the FDA and Biogen, the manufacturer, has allegedly been uncovered:
This report documents the atypical FDA review process and corporate greed that preceded FDA's controversial decision to grant accelerated approval to Aduhelm." Commerce Committee Chairman Frank Pallone, Jr. (D-NJ)
The questionable practices applied not only to the approval process but to the pricing (at $56,00 per year), which the Centers for Medicare & Medicaid Services (CMS) rejected.
Not two years later, Aduhelm’s manufacturer, Biogen, introduced another new drug into the market, this one called Leqembi. It received accelerated approval in January and “traditional” approval in July. This drug reduces plaques, which are associated with Alzheimer's in about 75% of cases (but are not necessarily causal), and also might improve cognition in patients with mild cognitive impairment (MCI). Alas, it has some serious side effects -- including death. The first clinical trial reported three deaths; another reported 13 deaths in a cohort of 1800 people. But there’s more: cerebral edema (swelling) and hemorrhage were also noted, and patients with a particular genetic signature (apolipoprotein E4-ApoEF) were considerably more likely to suffer these conditions while receiving no cognitive benefit. Nevertheless, package labeling does not require genetic testing prior to administration.
“In fact, the label even indicates that the drug can still be given if such genotyping is not performed" - Jerry Avorn, M.D
The outcomes for Leqembi are equivocal. While some improved cognitive results were seen in the cohort of 1795 participants, the issue remains whether the improvement was clinically meaningful. All participants started from a baseline of mild to very mild cognitive decline and worsened – and the drug didn’t stop cognitive decline, just slowed it.
For those skittish about using another Biogen drug, Eli Lilly has another drug in the wings and expects FDA approval by year-end. This one is called donanemab, which, like Leqembi, is a monoclonal antibody. While donanemab slowed the progression of Alzheimer's by about 35%, which sounds impressive, this only equals a seven-month respite in decline- and that’s only in those with mild cognitive decline. Donanemab, too, comes with serious side effects. Brain scans revealed side effects of brain swelling or hemorrhage in about 25% of patients. Here, too, patients died — three of them.
“[E]xperts caution that donanemab is no cure, and that its benefit amounts to only about a seven-month delay in the loss of memory and thinking.”
Mild Cognitive Impairment(MCI)
MCI is a decline in thinking and memory abilities. It’s not pathognomic or diagnostic of dementia, Alzheimer’s, or any other form. Often, it precedes it, but not always.
“MCI is a concerning condition due to the effects it can have on an individual’s quality of life, and it may also indicate the early stages of degenerative neurological diseases such as Alzheimer’s. At times there are also psychological impacts due to MCI that may lead to anxiety, depression, and other mental health disorders.”
- Psychiatrist, Dr. Simon Faynboym
So, what’s an old fart with waning memory to do?
Last week, a new study in Alzheimer’s and Dementia [1] reported on a new clinical trial yielding “significantly” improving memory and executive function with minimal or moderate side effects in an older population with mild cognitive decline. The Alzheimer’s Association touts the study as a success. Not surprisingly, they also championed the approval of Leqembi and Aduhelm, which it welcomed enthusiastically. But do the results bear out the group’s ecstasy?
A reduction in side effects is not surprising since the preparation, Sailuotong (SLT), is a Chinese herbal medicine containing Panax ginseng, Ginkgo biloba, and Crocus sativus in a 5:5:1 ratio. Given the drama incident to the other drugs, it is tempting to view this new offering either as snake oil or to run out and stock up [2]. So, first, here’s a deep dive into the “study” to see if it’s worth scouring the market.
Compared to the monoclonal antibody studies submitted to the FDA, all of which involved cohorts of about 1800 (half tested and half placebo), the STL study recruited 78 participants, 39 each randomized into treatment and control groups. Only the logical memory delayed recall component and executive function [3] yielded positive and statistically significant results.
To my mind, the cases and controls seemed lopsided concerning gender (59% female in the test group, 46% in the control and the use of psychoactive medications, 29% in the test group, and 44% in the control, making me question the randomization process (although in small studies, skewed data are legitimately possible). Further, given that depression impacts cognition, one wonders what effect either the underlying condition or the medications used by the control group had on the test results. [4]
There were two fascinating and cautionary findings. First, daily functionality improved in the control group! The researchers disclaimed the significance of that finding, saying the discordance wasn’t clinically meaningful. Second, significantly, serious adverse events occurred only in the controls! (This might mean it is hazardous to engage in clinical studies, and the cost-benefit isn’t viable).
Comparing the studies for the different drugs is impossible as the tests used to measure cognitive abilities differed, but biological plausibility for the Chinese extract exists. Past studies (mostly in rats, mice, and in vitro) report that Sailuotong has an anti-inflammatory, antioxidant effect, reduces platelet aggregation, enhances cholinergic function (of the neurotransmitter of acetylcholine), increases cerebral blood flow, and may reduce beta-amyloid plaques. It also has anti-depressant properties and reduces anxiety, all of which should aid in cognition – and mood. At least one small proof-of-concept study demonstrated cognitive benefits at one week.
Chicken Soup and Positive Thinking
I would place this Chinese extract in the “chicken soup” category – it might help, but it shouldn’t (seriously) hurt. The Ginseng-ginko formulations I could find (although not the formulation tested) were fairly inexpensive, certainly about a thousand times cheaper than the $26,000 yearly price tag affixed to Leqembi. The adverse effects were no more serious in the test group than in controls, mainly GI issues. There were no deaths.
On the other hand, there might yet be an even better and safer alternative. Mild cognitive impairment is believed to be a progressive decline. But that may not necessarily be the case. While the drugs tested are marketed to stem or slow the decline, it may well be that a “cure” is possible! It turns out that at least one study demonstrates that a positive attitude can actually reverse the decline.
A randomized clinical trial of over 1700 participants reported in JAMA Network six months ago found that attitude, perception, and stereotypes individuals hold about aging may impact outcomes: And those holding positive beliefs about aging registered better MCI results (recovery) and continued improving.
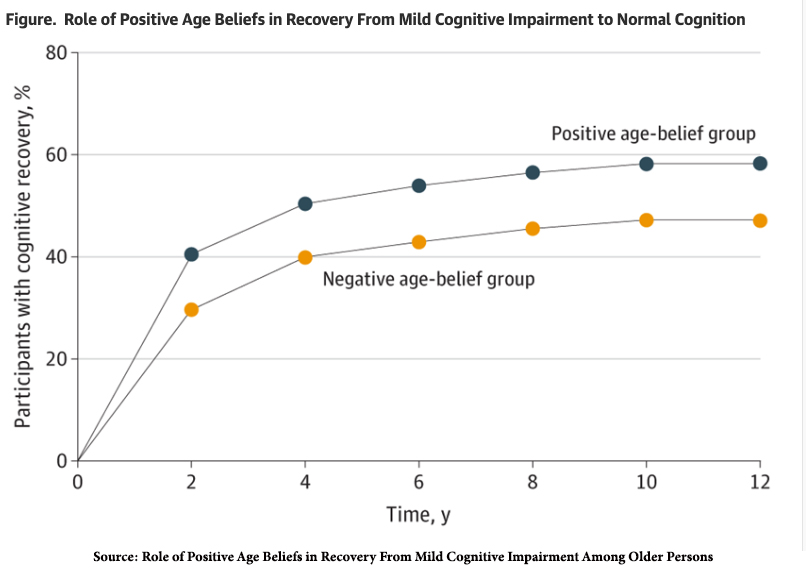
“Participants with MCI at baseline were significantly more likely to experience cognitive recovery if they had positive age beliefs at baseline…. The positive age-belief group had a 30.2% greater likelihood of recovery than the negative age-belief group; this recovery advantage persisted regardless of baseline MCI severity.”
Interestingly, almost half of the individuals with negative age beliefs also developed complete cognitive recovery. However, those with positive age beliefs reached that state of recovery faster and “were significantly less likely to develop MCI over the following 12 years compared with those with negative age belief,” meaning positive thinking could also be preventative.
Norman Vincent Peale told us years ago of the power of positive thinking. Who knows, it might also work for preventing age-related cognitive decline?
[1] The journal is funded by the Alzheimer’s Association, a non-profit patient advocacy group.
[2] I can’t find any on the web -although Panax ginseng and Ginkgo biloba combinations are available, albeit in a different formulation
[3] switching between cognitive concepts, higher-level divided attention, and multitasking.
[4] While the p-values of the differences were not statistically different, I believe these variables should have been controlled in reporting the results.




