I'm not a big fan of essential oils. They are simply complex mixtures of plant-derived chemicals called terpenoids (See Essential Oils - Not What The Name Implies). None of them are especially harmful and, with the exception of having a nice scent, they are not especially helpful either. But I happened upon an interesting paper from 2009, which claims that tea tree oil, the olfactory darling du jour, might be useful in treating herpes.
But before I tell you about why Paul Schnitzler and colleagues at the University of Heidelberg may have something interesting on their hands, first you're going to need a little virology lesson. Sorry, but this ain't all fun and games.
Here's how you grow viruses in the lab (Figure 1).
- You start with empty little dishes which are called wells.
- Each well is filled with a growth media - something in which the viruses will replicate. In the case of viruses, the growth media consists of living cells. Different viruses require different host cell types.
- You wait.
- The host cells form what is called a monolayer in the well.
- Then the virus you want to grow is added to each well.
- You wait some more.
- The virus grows. There are a number of different ways to measure how much virus has grown. One is visual - looking for plaques - small dots of viruses in the growth media (Figure 1).
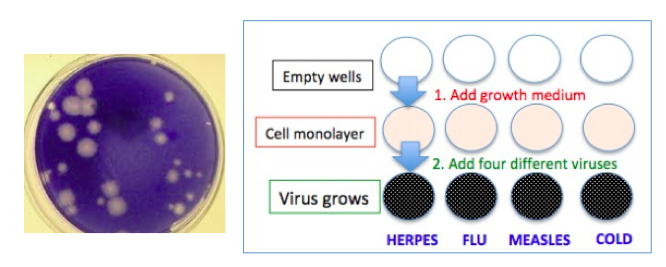
Figure 1 (Left) Viral plaques. Photo: Virology Blog. (Right) A simplified representation of growing viruses in a well. The darker the color, the more virus is present.
Now you can test a chemical, drug, etc. to see if it inhibits the growth of the virus in the cells. With a little luck this will be an indicator of whether the chemical/drug will work in a living body. Here's how it works.
EXPERIMENTS TO DETERMINE VIRAL INHIBITION OF 5 DRUGS
In Experiment 1, we are looking at the effect of Drug A on the replication (growth) of the same four viruses, herpes, flu, measles, and the cold virus, both in the absence and the presence of the drug being tested. As expected, when no drug is present (the control group), each virus grows without any problem (1). But when Drug A is added, there is no sign of growth in any of the viruses.

Experiment 1
Experiment 2 is identical to Experiment 1, but Drug B is used instead. Once again, the viruses all grow in the absence of B, but in this instance, the drug slightly inhibits the growth herpes, flu, and measles, but not the cold.

Experiment 2
In Experiment 3, Drug C stops the growth of herpes but does not affect the flu, measles or cold viruses.

Experiment 3
In Experiment 4, Drug D has no impact on any of the viruses.

Experiment 4
In Experiment 5, Drug E inhibits the growth of flu but not any of the others.

Experiment 5
Now it's time for a quiz! It's gonna be LOTS of fun! Good luck. You'll need it.
- Which of the five (A-E) is the most promising viable herpes drug?
- How many of the five could be considered to be viable herpes drugs?
- How many of the five could be considered to be viable drugs against any virus at all?
AND HERE ARE THE ANSWERS!!!

- Drug C
- One
- Two
THIS MAKES NO SENSE NOW. BUT IT WILL.
Unless you are up on your virology, this probably sounds pretty crazy right now. You may even be feeling a bit used, as if someone had played a sleazy trick on you. You would be right. Sleazy Josh has done just that. For a reason. You have just been introduced to the selectivity panel.
THE SELECTIVITY PANEL - KEY TO VIROLOGY RESEARCH
Viruses are obligate parasites, which means that they can only replicate in healthy living host cells. This means that any drug that either a) stops the virus, or b) kills or damages the host cell will appear to have antiviral properties. It is critical to differentiate between the two. One way to accomplish this is running a selectivity panel. Here's how it works.
When two viruses are very different, especially if they are from different families, it is extremely unlikely that they will contain the same internal machinery - enzymes or proteins - required for replication. So if a chemical shuts down both flu and herpes, which are dissimilar, the chances are very high that the chemical is simply poisoning the host cells rather than stopping a specific step in the virus inside the cells. In this way, unrelated viruses from different families are used as internal controls for each other to distinguish between toxic compounds and potential antiviral drugs. The five experiments above illustrate just that.
In Experiment 1, Drug A prevented replication of all four dissimilar viruses. Drug A is a toxin, not an antiviral drug. Bleach would produce the same result.
In Experiment 2, Drug B slightly inhibited replication of three dissimilar viruses but not the fourth. Drug B is a weak toxin, not an antiviral drug.
In Experiment 3, Drug C inhibited replication of herpes, but not any of the others. This means that Drug C is probably a direct-acting antiviral and is potentially useful against herpes.
In Experiment 4, Drug D did not prevent any viral replication. It is neither a toxin or an antiviral.
In Experiment 5, Drug E prevented replication of flu, but not any of the others. This means that Drug C could be a potential flu drug.
These five experiments demonstrate the primary method that researchers in virology use to tease out toxicity from antiviral activity. But there are other methods, one of which will be shown below.
WHAT DOES THIS HAVE TO DO WITH TEA TREE OIL?
Now (assuming you haven't choked to death on your own vomit) let's see how this applies to tea tree oil (Figure 2).
Figure 2. The effect of increasing concentrations of various essential oils on herpes infectivity.
Figure 2 shows the effect of 12 different essential oils - at different concentrations- on the ability of herpes to keep replicating. At the lowest concentration tested, 1 microgram per milliliter, only thyme (turquoise line) an citral (yellow) inhibit the virus, and only slightly. By the time the concentration reaches 10 micrograms per milliliter, 6 of the 12 oils show at least 50% inhibition. At 25 micrograms per milliliter (orange circle), tea tree oil (red line), alpha-terpinene (green), and alpha-pinene (blue) have completely eliminated infective herpes virus. Does this mean that these three are good drug candidates? No. Because we still cannot tell whether the effect being measured is due to cell toxicity or a real antiviral effect.
Figure 2 addresses this question, but by using a different method than I described above - comparing the concentration that kills the host cells to the concentration that inhibits the virus in these same cells. This type of selectivity index is less rigorous, but it serves to illustrate how virologists can tell whether they are chasing down a false lead (a toxin) or working with something real. Figure 2 makes it clear that tea tree oil is both the most potent essential oil (it inhibits half the viral growth at 2 micrograms per milliliter) and also the most selective. The difference between the amount of the oil to kill the cells and the amount needed to stop the virus is 60-fold (the selectivity index).
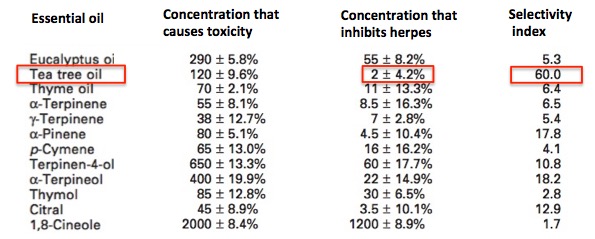
Figure 2. The selectivity indices of 12 essential oils. Concentrations are expressed in micrograms per milliliter. Tea tree oil (red boxes) is the most potent and most selective of all the essential oils tested. Figures 1 and 2 are adapted from "Comparative Study on the Antiviral Activity of Selected Monoterpenes Derived from Essential Oils." J. Phytother. Res. (2009)
DOES THIS MEAN THAT TEA TREE OIL WILL CURE HERPES?
No, it doesn't. For a number of reasons. Here are some of them.
- There is no cure for herpes, only drugs to control it.
- Tea tree oil is not very potent. Its EC50 - the concentration necessary to inhibit half the growth of the virus - is 2 micrograms per mL. Many drugs are far more potent; they have EC50 values that are thousands of times lower. The more potent a drug the less you need to take, which means better efficacy and fewer side effects.
- Although 60-fold selectivity may sound pretty good, it's not. For antiviral drugs, this number can be in the thousands. Even millions.
- Although in vitro cell culture experiments are very useful in determining whether a given compound will stop viral replication in a dish, they tell us nothing about how that same compound might function inside your body if given orally. Pharmacokinetic parameters, such as metabolism, toxicity, half-life, others, are the biggest hurdle that most drugs will face.
- It is far more likely that a topical application would be effective than a capsule, but that does not mean that it is strong enough to do so.
- Tea tree oil is not a single chemical; it is a complex mixture of many chemicals. It is plausible, if not likely, that only one of the chemicals is responsible for the antiviral effect. The other chemicals greatly increase the chance of toxicity, especially if the oil is to be given orally in a dose high enough to inhibit viral replication.
- This paper was from 2009. If tea tree oil was effective in animal models of infection and wasn't overtly toxic, you can bet the ranch that we would know about it.
This article really isn't about whether tea tree oil is an effective drug in treating herpes. If it is, it is probably only modestly useful. Rather, it's about one piece of a very complex puzzle of how antiviral drugs are discovered. And doing this is anything but easy.
From the time of the discovery of HIV, it took 15 years for the first direct antiviral drug to be approved. For hepatitis C, it was 22 years. Even though these drugs would ultimately succeed, getting them to patients still took forever because of the many hurdles that drugs must overcome to make it from the lab to the pharmacy.
Potency and selectivity are only two of these hurdles.
Congratulations. If you made it through the end of this thing you are now a "virologist."
(NOTES)
(1) Different viruses require different cell types of cells in which to grow. Herpes grows in human foreskin cells (this should not be surprising) called fibroblasts.




