
Recent rumors speculate that ivermectin and Pfizer's promising experimental Covid drug PF-07321332 (1) are both inhibitors of the viral main protease (Mpro) (2) and can therefore be used interchangeably to treat Covid infections. It's gotten so silly that a new term has been invented – "Pfizermectin," implying that Pfizer's Paxlovid is little more than an expensive, big-pharma ripoff of ivermectin. This rumor really has legs, so let's examine whether there is anything to it. Are ivermectin and PF-07321332, more commonly known as Paxlovid the same? Similar? (Note: The active component of Paxlovid is PF-07321332; another drug called ritonavir is added to prolong its half-life.) No, they are not.
To be able to compare ivermectin and PF-07321332 we need to look at two pharmacological measurements. One is the IC50– the half-maximal inhibitory concentration, which is the concentration at which either a biological process (such as viral replication), or enzymatic reaction within a cell, is inhibited by half. The other is Cmax – a measure of the highest attainable blood levels of a drug after it is administered. They are intimately connected.
IC50 is sometimes called the EC50 (effective concentration 50%). They are similar, but not identical. (3). When IC50 /EC50 values are high it means that more drug will be required to inhibit the process in question (a low potency drug). Conversely, when the IC50 is low it means that the drug is more potent and less drug will be needed. Good drugs generally have low IC50 values. It's one of the most important parameters in antiviral drug discovery – something I did for 10 years.
Below is a table which can be used as a rough, descriptive guide of a range of potencies and the probability of whether a compound of a given potency has a reasonable chance of being "strong" enough to be a useful drug. Values are approximate:
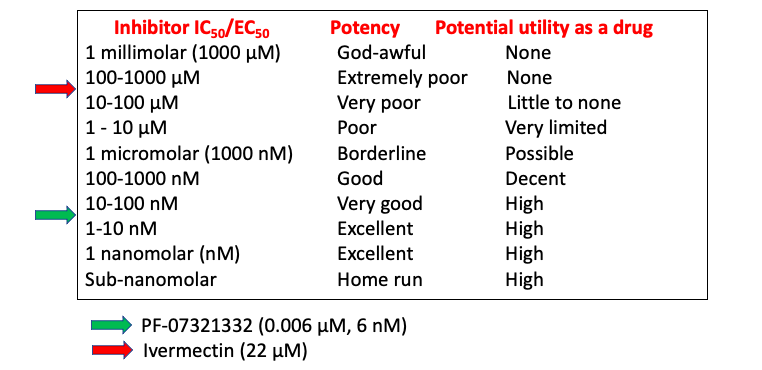
Table 1: The approximate IC50 values vs. Mpro of ivermectin (red arrow) and PF-07321332 (green arrow) are shown.
Ivermectin vs, PF-07321332 as inhibitors of the Mpro enzyme?
- Ivermectin
Taval et. al. studied a group of chymotrypsin-like compounds as potential inhibitors of Mpro. The group reported in the journal Communications Biology that:
"ivermectin was able to inhibit more than 85% (almost completely) of 3CLpro (another name for Mpro) activity in our in vitro enzymatic assay..."
While this may sound impressive to the uninitiated, it is not. This is because in order to attain 85% inhibition of the Mpro enzyme a concentration of 50 µM (micromolar) of ivermectin was required – an absurdly high number for a potential drug. There are probably hundreds, if not thousands, of chemicals in the world that will inhibit Mpro (and plenty of other enzymes) at this concentration.
"...with an IC50 value of 21 µM"
A value this high is normally the kiss of death for a drug. Table 1 shows just this. Inhibitors with IC50 values in this range are just not potent enough to be useful drugs.
- PF-07321332
Although Owen et.al. did not indicate the IC50 of PF-07321332 against Mpro in their recent Science paper, the authors did report a binding constant (Ki) of 3.1 nM. There is a difference between the two and I'm BEGGING you not to ask me to explain it. Suffice it to say that it has been estimated that for most inhibitors, the IC50 is twice the Ki, making it something like 6 nM, making PF-0732133 3,500-fold more potent than ivermectin in this assay. From these values it can be concluded that PF-07321332 is a potent inhibitor of Mpro while ivermectin barely inhibits the enzyme at all. Whatever antiviral properties ivermectin may have cannot be a result of its inhibition of Mpro.
Ivermectin vs. PF-07321332 as inhibitors of viral replication in cultured cells
Cell-based (in vitro) assays are critical in developing antiviral (and other) drugs because they demonstrate whether the drug in question can pass through a cell membrane, perform its specified function, and inhibit the replication of virus within the cell. The primary drawback of cell-based assays is they tell us nothing about how the drug works (what the molecular target is). For example, Merck's molnupiravir will inhibit the growth of SARS-CoV-2 in cells but will show no activity in the Mpro assay because it operates by inhibiting a different viral target.
- Ivermectin (IVM)
Caly and colleagues reported that in a cell-based assay, ivermectin inhibited viral replication (as measured by the production of viral RNA) with an IC50 of ~2 µM, quite a high concentration. Can this concentration be achieved in blood following administration of a safe dose of the drug? A paper by Schmith and colleagues titled "The Approved Dose of Ivermectin Alone is not the Ideal Dose for the Treatment of COVID-19" (4) concluded no – it cannot. Some highlights include:
At the approved ivermectin dose (200 micrograms per kilogram of body weight) –12 mg for a 60 kilogram human – the maximum concentration (Cmax) of IVM in the blood was 47 nanograms/mL. When compared to the IC50 of IVM (2 µM, 1,750 ng/mL) the Cmax is far lower than the IC50 (by 35-fold). In other words, there is not nearly enough drug in the blood to inhibit half of the viral replication.
Neither higher doses – 120 mg IVM (10X the recommended dose) nor multiple doses of 60 mg provided a substantial increase in the Cmax. It is safe to conclude that IVM is not potent enough to provide a concentration in blood that even approaches the drug's antiviral IC50 value, so whatever effect IVM on may be having on Covid it is not because of its antiviral properties.
- PF-07321332
We see a very different profile for Pfizer's drug (Figure 1)
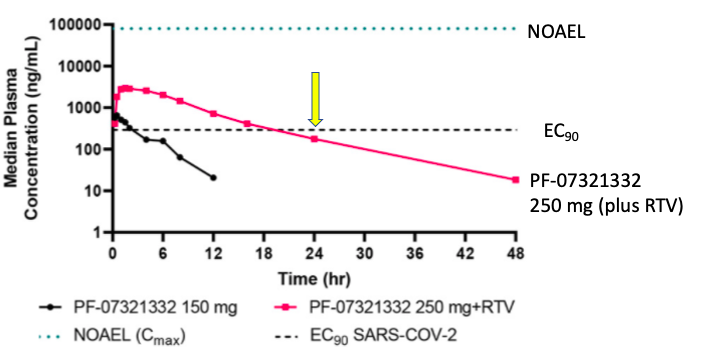
Figure 1. A single dose of PF-07321332 (250 mg plus ritonavir, pink line) provides blood levels at or above the drug's EC90 (black hatch line) – the concentration of the drug that inhibits 90% of replication — a much more stringent measurement of viral inhibition – for 24 hours (yellow arrow) while remaining far below the no-observed-adverse-effect level (NOAEL, blue dotted line) – the blood level at which no drug-related side effects are observed. This is the type of profile expected of a antiviral drug.
Source: Science
Bottom line
- It is abundantly clear that the "Pfizermectin" myth is just that. PF-07321332 is a potent enzymatic inhibitor of Mpro with proven efficacy in clinical trials. Ivermectin has very poor potency in this same assay. Ivermectin, if it works at all, is certainly not functioning as a protease inhibitor.
- Based on in vitro studies, Ivermectin is a weak inhibitor of viral replication in cultured cells.
- But the antiviral inhibition of ivermectin is weak enough that even at high or multiple doses of the drug blood concentrations do not reach its cellular assay IC50 of 2µM.
- PF-07321332 is a potent inhibitor in both the enzymatic and call-based assays.
- The drug reaches and remains at blood concentrations sufficient to inhibit 90% of viral replication in cells for one day and does so well below toxic levels.
These data tell us that ivermectin neither inhibits the viral protease in an enzyme-based assay nor slows the production of virus in cells while Pfizer's drug does both. If ivermectin is proven to be an effective Covid treatment it must be acting by some yet-unknown mechanism.
NOTES:
(1) PF-07321332 is sometimes used interchangeably with Paxlovid, the trade name of the drug. This is mostly accurate but not entirely. Paxlovid contains the active ingredient PF-07321332 in combination with a small amount of a second drug called ritonavir, which improves the blood levels of PF-07321332 by inhibiting the enzyme that metabolizes it.
(2) For more information on Mpro see Pfizer's COVID Drug Works Wonders. Here's How It Works.
(3) There is a very similar parameter used to measure inhibition called an EC50, which stands for effective concentration rather than inhibitory concentration. They are often used interchangeably. Let's play "don't ask, don't tell." You don't ask and I won't tell.
(4) Clinical Pharmacology & Therapeutics, Volume 108 #4, October 2020. doi:10.1002/cpt.1889



