Wow! According to a new paper in the Journal of Natural Products, two chemicals found in marijuana could be helpful in the prevention of COVID infection, something that isn't enormously surprising; that some component of marijuana would be touted as a weapon against COVID was pretty much a foregone conclusion. After all, since the decriminalization of marijuana, we have been inundated with claims of the benefits of cannabis. CBD oil is (supposedly) helpful in treating no fewer than 15 conditions, ranging from MS to cancer.
Now, Richard B. van Breemen, PhD at the Linus Pauling Institute, and colleagues have reported that two chemicals from hemp extracts – cannabigerolic acid (CBGA) and cannabidiolic acid (CBDA) bind to the viral spikes blocking the COVID virus from entering the host cell. CBGA and CBDA are known to be orally bioavailable and safe; there is also historical precedent for drugs employing this same approach being successful against HIV:
The approach of utilizing compounds that block virus–receptor interaction has also been useful for other viruses, including HIV-1 and hepatitis C virus.
On the surface, everything looks great. Why wouldn't these two compounds make viable COVID drugs? But, if you look a little deeper, it's not so simple. First, let's look at the historical precedent:
- Although drugs that block cell entry are being investigated as potential therapies for hepatitis C, there are no approved drugs for this purpose.
- Fuzeon is an AIDS drug that belongs to the cell entry inhibitor class, and it does indeed prevent viral entry into host cells. Fuzeon is important in treating people who have failed first-line therapies.
So what's the problem with the cannabis compounds?
It's all about potency. Antiviral drugs will block viral replication, but only potent drugs will be able to do so at a blood level that is attainable. That's where the problem lies. The cannabis compounds aren't very potent, so it would take a very high, maybe impossibly high, blood concentration for them to adequately stop COVID replication.

Source: Antimicrobial and Chemotherapy 54(1):134-42
The IC50 is the concentration required to inhibit 50% of viral replication in cells; the lower the number, the more potent the inhibitor. For CBDA against the original Wuhan strain and two early variants, the IC50 ranges from 11-24 micrograms/mL, which equals 31-67 micromolar. The potency of CBGA is a bit less: 73-104 micromolar. But, what do these numbers mean? To put the data in perspective we need to look at the cell-based potency of other antiviral drugs (Table 1).
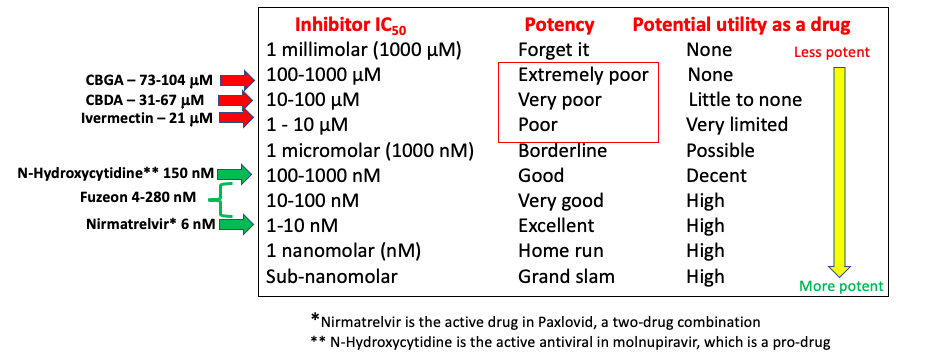
There's a lot going on in Table 1. Let's compare the two cannabis compounds with other drugs that inhibit COVID replication.
- The yellow arrow and the column next to it give a rough approximation of the chances of compound being potent enough to possibly be a drug.
- The "distance" between the compounds represented by the green arrows and those with red arrows is immense; this speaks directly to the relative antiviral potency of the compounds in the two groups.
- While CBGA, CBDA, and Fuzeon (HIV) all act as cell entry inhibitors, Fuzeon, with IC50 values ranging from 4-280 nanomolar (1), is 300-20,000 times more potent in cell-based assays than CBGA and CBDA. In other words, at low blood concentrations, Fuzeon will efficiently block HIV replication while CBGA and CBDA will not do the same with COVID.
- The antiviral potency of ivermectin is similar to that of the cannabis compounds and very different from the three approved antivirals. (2)
- Nirmatrelvir, the antiviral component of Paxlovid, is a highly potent anti-COVID drug with an IC50 of 6 nM.
- N-Hydroxycytidine, the active form of molnupiravir (a pro-drug) has an IC50 of 150 nM making it about 25-times less potent than nirmatrelvir, but still well within the range of known antiviral drugs. This difference in cell-based potency between molnupiravir and nirmatrelvir may very account for the clinical superiority of Paxlovid.
Bottom line
It is not a coincidence that the drugs represented in green (low IC50 values) are approved antiviral drugs while the drugs in the red area are not (and probably will never be). Antiviral drug discovery is very challenging, but it's not magic. Antiviral drugs must inhibit the replication of the virus within living cells, or they cannot be expected to do so within the bodies of animals or humans. Unfortunately, the cannabis compounds are such weak inhibitors that the chances of being therapeutically useful are slim.
NOTES:
(1) The significant range in the potency of Fuzeon is due to the large numbers of HIV strains that it was tested against.
(2) I recently wrote an article comparing the antiviral potency of ivermectin and Paxlovid in which I demonstrated that ivermectin was not an effective direct-acting antiviral drug. As for the discussion of whether ivermectin actually works or not, as the great Yogi Berra once said, "include me out."




