Multiple sclerosis is an autoimmune disease that primarily affects the myelin sheaths (white matter), which enhance nerve conduction and surround the nerves. For most MS patients, there are relapsing episodes, and radiologically, MRI studies show areas of acute and chronic demyelination. These areas can be seen incidentally on MRI studies before the onset of MS and are termed “radiologically isolated syndrome.” Their presence points to pathological changes present long before they become clinically apparent – MS has an early, subclinical phase.
Other immune diseases, like rheumatoid arthritis, share a subclinical, prodromal phase, and research has identified and characterized diagnostic autoantibodies for them. The current study sought an autoantibody signature for MS.
B-cell depletion
B cells make antibodies and do not normally cross the blood-brain barrier. In MS, B cells have been found to have entered the central nervous system and attack the myelin sheath surrounding your nerves – demyelination. Monoclonal antibodies directed at these B cells have been shown to slow the demyelination by “reducing inflammation.” A beneficial side effect is that the reduction in B cells reduces inflammation further by increasing the presence of regulatory T cells and reducing the number of T cells recruited to the areas of inflammation - proteins that fight viruses and bacteria. B cells usually can’t cross from your blood into your brain or spinal cord. If you have MS, some B cells enter your brain and spinal cord, attacking the myelin sheath that protects your nerves. Of course, the reduction in immune cells increases the risk of infection, and one in particular, progressive multifocal leukoencephalopathy PML), is a rare brain infection associated with B-cell depletion therapy.
The researchers made use of the Department of Defense Serum Repository established in 1989 when the Department of Defense mandated HIV testing of all members of the military. It now contains serial serum specimens from 12 million service men and women. A subset of the repository taken from Gulf War veterans with S provided the dataset. Patients with MS had serum samples from 5 years before and one year after the clinical onset of MS; controls were drawn from age, gender, and race matched members of the cohort – a total of 250 individuals overall.
Findings:
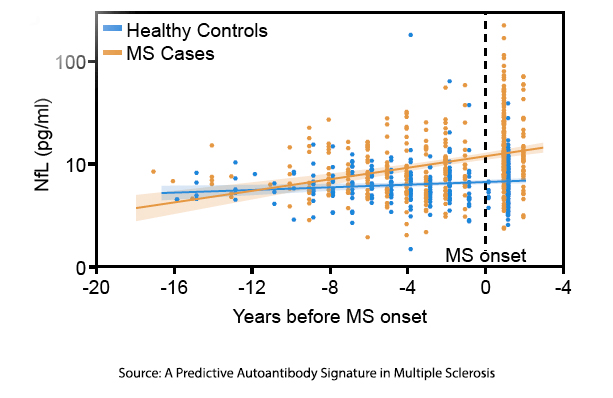 Neurofilament light chain (NfL) – this is a protein found in neurons and considered a biomarker of neuronal damage in MS. It is released into the cerebrospinal fluid and then into the blood, where it is termed sNfL and can be seen up to six years before MS presents clinically. Researchers found that patients with MS developed increasing sNfL titers over time, continuing to rise after MS became symptomatic, while there was no change in the titers of sNfL in the controls.
Neurofilament light chain (NfL) – this is a protein found in neurons and considered a biomarker of neuronal damage in MS. It is released into the cerebrospinal fluid and then into the blood, where it is termed sNfL and can be seen up to six years before MS presents clinically. Researchers found that patients with MS developed increasing sNfL titers over time, continuing to rise after MS became symptomatic, while there was no change in the titers of sNfL in the controls.
The researchers then identified a cluster of antibodies against a unique group of proteins, an “antibody signature” for about 10% of individuals developing MS with early increases in sNfL. The signature remained stable over time, was highly specific for MS rather than other neurologic degenerative diseases, and, more interesting, those included proteins were associated with several human pathogens, including Epstein-Barr virus (EBV). Infection with EBV, in another study, was found to increase the risk of MS 32-fold. [1]
The identified autoantibodies might contribute to the development of MS by
- Directly targeting antigens on myelin
- Mistake epitopes, the area to which an antibody attaches, of the EV virus or other microbes with areas on neuronal tissue (termed molecular mimicry)
- Cause an inflammatory response to something other than myelin or neuronal material where white matter is a “bystander,” caught up in the inflammatory reaction with disruption of function or demyelination.
Their findings allowed the researchers to conclude support for
“the notion that neurodegeneration is already occurring as a result of underlying immune-mediated neuroaxonal pathology in the prodromal phase of MS.”
This autoantibody profile is present before, during, and after the first symptom onset, suggesting its potential utility as a biomarker for early disease detection and monitoring. There currently is no cure for MS, and the most critical therapeutic interventions are disease-modifying therapies like the B cell depletion mentioned earlier.
There are currently multiple FDA-approved disease-modifying therapies for MS, targeting eight distinct areas of the immune-mediated disease process. They are effective to varying degrees in slowing the clinical symptoms of the predominant form of MS, relapsing-remitting MS, but there is no approved therapy for the 3% who develop primary-progressive MS. Finally, these therapies are expensive, with an estimated cost by the National MS Society of
“$57,202 to $92,719, depending on age and gender. In February 2022, the median annual price of a brand-name DMT was close to $94,000. The average wholesale cost of an oral agent is about $8,000 to $90,000 a year.”
In conclusion, the recent advancements in understanding the autoimmune warfare underlying multiple sclerosis (MS) represent a significant leap forward in both diagnosis and treatment strategies for this complex condition. By identifying a predictive autoantibody signature and recognizing neurodegeneration occurring in the prodromal phase, researchers offer hope for earlier detection and monitoring of MS progression. As we move forward, this newfound understanding may pave the way for more targeted and practical approaches to managing the impact of MS on the lives of millions worldwide.
[1] Other pathogens included Hepatitis C, Pseudomonas, E. coli, and Aspergillus.
Source: A Predictive Autoantibody Signature in Multiple Sclerosis Nature Medicine, DOI: 10.1038/s41591-024-02938-3




