News outlets just love to scare people. Scares get readers. Readers mean money. Most of the time the scares are overblown. Not this time. There are currently a whole bunch of reports about Giant Hogweed (which could fairly be called "Satan's Favorite Botanical") being spotted in a number of states, most recently Virginia. I recently wrote about the toxin in poison ivy (See Urushiol, The Poison In Poison Ivy). The chemicals in Giant Hogweed make poison ivy look like iceberg lettuce. Yes, they are that bad.
The news stories tell you to stay the hell away from the stuff. Some show photos of what can happen to skin that is exposed to furanocoumarins, the class of chemicals that is responsible its toxicity, especially if the skin is exposed to any sunlight (Warning: not for those who are easily grossed out). This is called phototoxicity.
What is not in the news is what makes this story especially interesting. Within the class of toxic chemicals that make the plant so dangerous (1) is an approved drug called psoralen. Psoralen is used in conjunction with UVA radiation (the combination is called PUVA) to treat psoriasis. The mechanism by which psoralen treats psoriasis is identical to that which causes the plant to be so toxic - damaging DNA. Given this, it is not surprising that PUVA is known to cause skin cancer, which makes this therapy less than ideal. There is a very large ongoing research effort to find better therapies for psoriasis, which when severe, can ruin the lives of those afflicted by the disease.
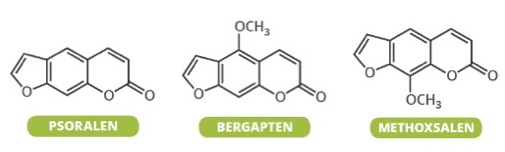
Figure 1. Some of the chemicals found in Giant Hogweed. Note the structural similarity between the three. They all work by the same mechanism.
INTERCALATION - A GREAT WAY TO SCREW UP DNA
Psoralen, which is found in citrus fruits and celery, not only disables the skin's ability to protect itself from UV radiation but also actively damages it. This is because it is perfectly designed to mess up your DNA. It does so by inserting itself into the grooves of DNA. Drugs and chemicals that do this are called intercalating agents. Once intercalation occurs (Figure 2) the DNA is damaged and can no longer perform its primary function - to make proteins. This is why intercalating agents are toxic.
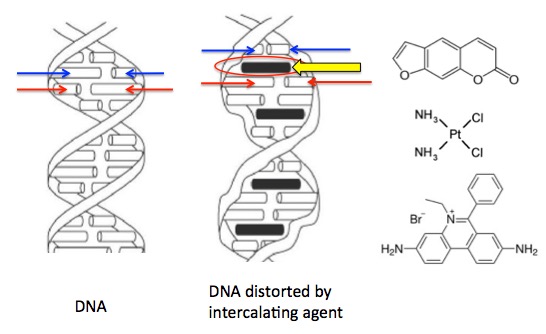
Figure 2. (Left) DNA consists of two strands, each consisting of a backbone (ribbon) of phosphate bonds that are bound to base pairs (red, blue arrows). The base pairs (there are four of them) hold together the single strands. A set of three consecutive base pairs (A,C,T,G) determines what amino acid will be incorporated into the protein that is being built by the DNA. This is called the genetic code. (Center) Intercalating agents (yellow arrow) fit tightly between two base pairs and distort (damage) the DNA strand. (Right) Three intercalating agents. They are all flat, which enables them to fit between the base pairs. Top: psoralen, middle: cisplatin, bottom: ethidium bromide. All are toxic carcinogens. Source: What-when-how
FURANOCOUMARINS TAKE IT TO THE NEXT LEVEL BY REACTING WITH UV LIGHT: CROSS-LINKING
All of the three intercalating agents in Figure 1 are toxic and mutagenic, but furanocoumarins, psoralen being one of a family of natural products, hit your skin cells with a double whammy. They not only intercalate but then, because of a highly specific chemical structure, react with light. In doing so, they form a short-lived, highly energy form of the chemical (aka reactive species), which not only damages DNA by intercalation but then something even worse when once exposed to light - cross-linking (Figure 3). (2)
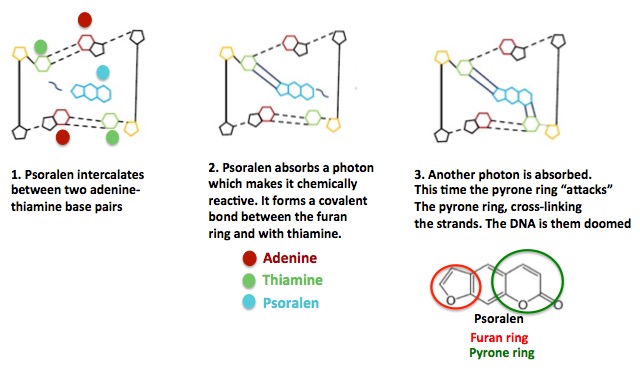
Figure 3. The mechanism of psoralen-based phototoxicity. (Left) Psoralen (and other members of the furanocoumarin family) penetrates the skin and get absorbed into cells. Then, because of their shape and chemical properties, the furanocoumarins fit between a specific pair of DNA base pairs. (Middle) The furanocoumarin absorbs UV light (this is rare in chemistry). The energy from the UV transforms the molecule from a low to high energy form. The high energy form is chemically reactive and forms a stable covalent bond with a nearby thiamine molecule. (Right) The furanocoumarin then absorbs more UV light, which again changes to a high energy species (a different form), which reacts with another thiamine molecule on the opposite DNA strand. This cross-links the two strands, making it more difficult for enzymes that repair damaged DNA to do their job. A number of chemo drugs work by cross-linking DNA strands.
So, if you haven't gone into a coma reading this thing you now know a bit about the science of why the plant is so dangerous. Feel free add it to your personal list of things to avoid. Here are two suggestions:

And another, which is even worse.

NOTES:
(1) In addition to a serious skin condition, eye exposure to furanocoumarins can cause blindness.
(2) DNA gets damaged constantly, but our cells have mechanisms to repair the damage. Damage from cross-linking is more difficult to repair.




