One of the strangest places on the planet (1), Lake Hillier, has been in the news lately. The lake looks like it could be a Pepto Bismol factory, but it's not Pepto or pollution; the color comes from bacteria that evolved colors for a specific reason – protection against the sun. Another relevant tidbit: Lake Hillier is VERY salty. How salty? Ten times that of the ocean.
A recent New Scientist article by Alice Klein described how scientists using DNA sequencing determined that some of the microorganisms in the lake color were novel; they had never been encountered. This brought up all sorts of questions. Why the color? How could anything live in such a salty environment? What purpose is the color? Is it from the salinity or some adaptive mechanism that enables the bacteria to live in such a strange environment?
Microbes that thrive in extremely salty water are called halophiles. There are not a whole lot of them on the planet, which was one of the reasons that the science behind their survival is so fascinating. It turns out that the salinity and the color are related, and (provided I can pull this off) it makes a nice story about how evolution/adaptation can work in strange ways to enable life to exist where it wouldn't be expected.
To truly understand the pink color, we sent Steve and Irving, our resident hosts for all Dreaded Chemistry Lessons from Hell © presentations from down under to... down under. This is very exciting! After all, Steve and Irving have been doing pretty much the same thing in the Eternal Hot Zone for millennia. They don't get out much.
Steve and Irving go to Australia!

A fairly common chemical reaction used by synthetic organic chemists use is called a [2+2] photocycloaddition. It involves irradiating molecules with carbon double bonds (alkenes aka olefins) with UV light; creating a rather unique reaction:
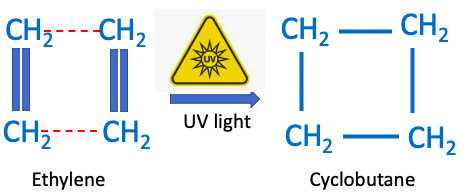
Figure 1. The [2 + 2] photocycloaddition reaction of 2 molecules of ethylene in the presence of UV light gives one molecule of cyclobutane. The red hatched lines show the new bonds formed.
What the hell does this have to do with pink lakes?
Everything. One of the DNA nucleobases, thymine, contains a carbon-carbon double bond in the molecule. The reaction is fundamentally the same as above: two alkenes reacting with UV light to form a 4-membered (cyclobutane) ring (red circle).
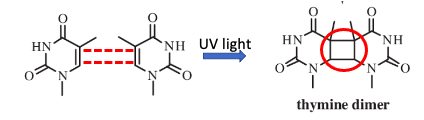
Figure 2. The [2 + 2] photocycloaddition reaction of 2 molecules of thymine forms what is called a thymine dimer. Note that the cyclobutane ring is present, just like in Figure 1.
One way to promote chemical reactions (this is one of the functions of enzymes) is to hold the two reacting molecules within bonding distance of each other (decreased entropy). Guess the perfect way to do this in vivo? DNA, but only when two thymine nucleobases are next to each other. Like this:
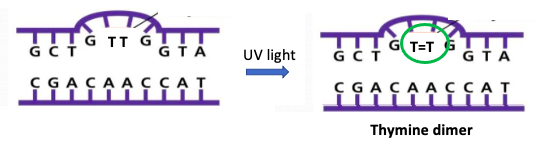
Figure 3. (Left) A fragment of DNA with two adjacent thymine nucleobases. (Right) When hit with UV light, the two thymine nucleobases undergo [2+2] photocycloaddition reaction causing a permanent (2) covalent bond (green circle). The reaction is identical to that in Figure 2.
What about the pink lakes?
This is where things get really cool. Below is a chart of the electromagnetic spectrum. The energy of a wave gets stronger going from right to left as its wavelength gets shorter.
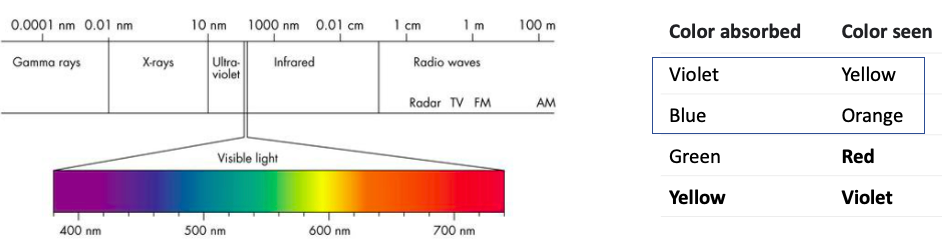
Figure 4. (Left) The electromagnetic spectrum. Humans can only see a sliver of the range of radiation (expanded color section). Note that violet and blue have the shortest wavelengths, therefore the highest energy frequencies, and are closest to UV. (Right) Chart of colors seen as a function of the color absorbed. Source: Coe.edu
The strange organisms in Lake Hillier have evolved to protect their DNA from sunlight by developing red, yellow, and orange (the combination of which looks pink) pigments that absorb UV light, preventing thymine dimers from forming and destroying strands of DNA. The pink color is simply a biological sunscreen.
What does the salt have to do with this?
The reason that pink lakes are highly salty is not intuitively obvious. After all, there are 75 bazillion living entities waltzing around in the sun (on both land and sea), and we don't look all like Pepto Bismol. So, the salt must have something to do with it, but what? A (very) little-known fact is that high salinity enables UV light to penetrate deeper than it does in fresh or less salty water.
Now it adds up. In an environment that would seem to be incompatible with life, certain specialized microorganisms evolved the ability to thrive where nothing else can survive (3), all because they learned how to defeat a chemical reaction that would otherwise screw up their DNA – a great example of the intersection of chemistry and biology.
Do Steve and Irving agree?

Not really...
NOTES:
(1) Lake Hillier is not the only pink body of water on the planet. There are about 30 others.
(2) The covalent bond on its own is permanent, but cells have complex repair mechanisms that undo the dimerization reaction. Don't even think about trying to read this reference. Your head will explode.
(3) Halophiles also have osmotic mechanisms enabling them to survive in highly salty water. These are beyond the scope of this article. And my intellect.




