As I have written many times, the laws and regulations that govern dietary supplements are a bad joke. But the joke is profitable - $40 billion per year from marketing untested drugs, vitamins that most people don't need, and mostly useless (and sometimes harmful) "dietary supplements," (1) complete with a sleazy disclaimer to make the sale "legal."
The industry blossomed from $4 billion per year to ten-times that after the passage of the scientifically bereft Dietary Supplement Health and Education Act of 1994 (DSHEA). It should come of no surprise that the law was championed by Senators Orrin Hatch (R-Utah) and Tom Harkin (D-Iowa). Utah is the home of many supplement companies, and just by sheer coincidence, Hatch is the #1 recipient of donations from the dietary supplement industry. Harkin is just an alternative medicine nut. Wouldn't surprise me if he has goat heads in his closet.
If you're not convinced, perhaps this will do the trick. Alzheimer's dementia is arguably the most devastating disease of all, especially for family members who will see a loved one slowly and inexorably decline into a person who has lost virtually all mental capacity. Not only is there no cure, but, after many years of study and numerous hypotheses, we don't have the wildest idea of what causes it. Although there are drugs that supposedly delay the decline, they are marginally helpful (at best) and have a boatload of side effects. In short, nothing works.
Here is a common fallacy: If "conventional" medicine doesn't have an answer for a particular disease or condition, then "alternative medicine" does. This is, of course, nonsense, but not to the supplement industry. They are only too happy to have the public delude "it$elf" in this way.
Which is exactly what happened recently. The FDA sent warning letters to companies that were selling thoroughly worthless supplements to "treat" Alzheimer's. Why is "treat" in quotes? Here's why:
Although supplement makers imply that their products are useful for treating a disease or condition by including the intentionally obfuscating term "supports" (a term that most people will equate with "treats" or "cures") on the label (in BIG) letters, the bottle must also contain a disclaimer on the label, which is in very small letters. Since it takes a scanning electron microscope to read the disclaimer on the label I've taken the liberty of resizing it:
"The disclaimer must also state that the dietary supplement product is not intended to "diagnose, treat, cure or prevent any disease,' because only a drug can legally make such a claim.'"
And how can there possibly be a better place to go to in order to get a rough idea of the relative size of the health claim and disclaimer? Why, it's Joe (Crazy Joe) Mercola's "Little Webshop of Horrors," which despite the nickname I gave it, is far from little. Here's one of many examples:

Image: Mercola.com
According to the FDA:
This is the essence of DSHEA - double talk. It's OK to make a "claim" on the label provided that you also put a disclaimer which pretty much contradicts the claim, assuming you can even read it. Nice.
Jonathan Jackson, a neuroscientist at Harvard Medical School really nails it:
"These anti-establishment, spunky, slickly marketed supplements seem to take care of the problem without the veneer of science.”
Jonathan Jackson, Ph.D, Popular Science 2/12/19
Wish I'd written that.
Back to the Alzheimer's scam.
So, what kind of useless crap was being peddled to the most helpless of all patients and their families by 16 supplement companies? The same old useless crap - mega-doses of vitamins, amino acids, minerals, etc. Here's one of the supplements cited by the FDA. Memory reVITALIZER, which was marketed by Alzheimer's Corporation - a COMPANY (with an intentionally misleading name), NOT an official organization (2) - for a mere $69.99 (but free shipping!).

Memory reVitalizer, one of the supplements that drew the wrath of the FDA. Photo: Amazon.com
There sure is a lot of stuff in there, right? Surely some of it must work, no?
No.
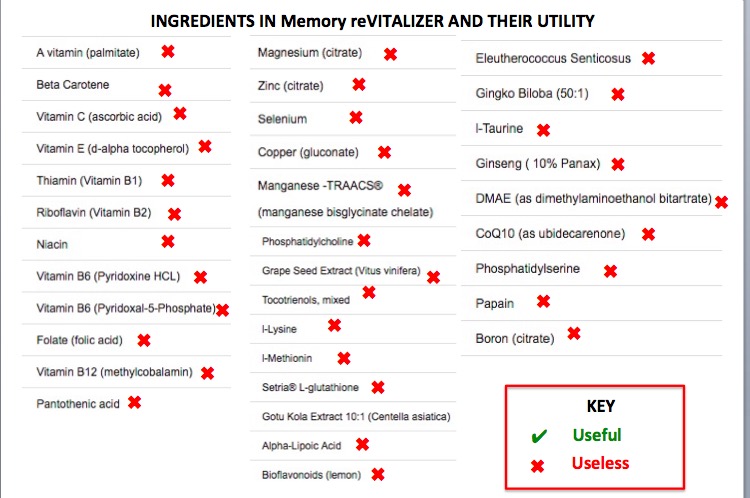
Ingredients in Memory reVitalizer and their utility in treating Alzheimer's
Although these products are worthless we need to give these companies credit for figuring out how to sell them. Dr. Jackson again:
“When a snake oil salesman gets called out, they just get a little more slick with their advertising.”
Ouch.
In the end, this latest incident is just more of the same. Dr. Scott Gottlieb, head of the FDA, who issued this press release should be commended for his statement in a recent press announcement where he says:
"One of my top goals is ensuring that we achieve the right balance between preserving consumers’ access to lawful supplements, while still upholding our solemn obligation to protect the public from unsafe and unlawful products, and holding accountable those actors who are unable or unwilling to comply with the requirements of the law."
But it's not going to be so easy. Gottlieb has a $40 billion albatross around his neck, thanks to Hatch, Harkin, and the magnificent sales (con) job that the industry has pulled over our eyes since 1994.
Let's wish him well.
NOTES:
(1) One of the "dietary supplements" that for some inexplicable reason, is allowed to be sold in drug stores and GNC, is dehydroepiandrosterone (DHEA), an anabolic steroid. Please explain to me how our diet fails to provide enough of an anabolic steroid, which can then be "supplemented." Supplement? My arse.
(2) How can these guys sleep at night? Pretending to be a patient advocacy or support group or medical organization? That is SO wrong. Here are a couple of real sites. WWW.alz.org, alzfdn.org. 
Image: Tenor.com




