Paxlovid, the “Tamiflu” of COVID-19, the oral medication shown to reduce hospitalizations, also is known for its “rebound” – a recurrence of COVID-19 a few days after completing the entire course of treatment. The New England Journal of Medicine reports on why – it’s the viral load.
The data comes from a double-blind, randomized, controlled trial of unvaccinated, symptomatic patients, PCR positive for COVID-19 and treated with Nirmatrelvir-Ritonavir (Paxlovid) within five days of symptoms – 2246 patients. [1] At the end of one month
- Paxlovid reduced hospitalization by 88% compared to controls.
- There were no deaths in the Paxlovid group and 13 in the placebo control arm.
Viral loads were measured at baseline and at least once during the follow-up period. Roughly 80% in each group, Paxlovid and placebo, had viral loads measured on Day 5 and during the “rebound period.”
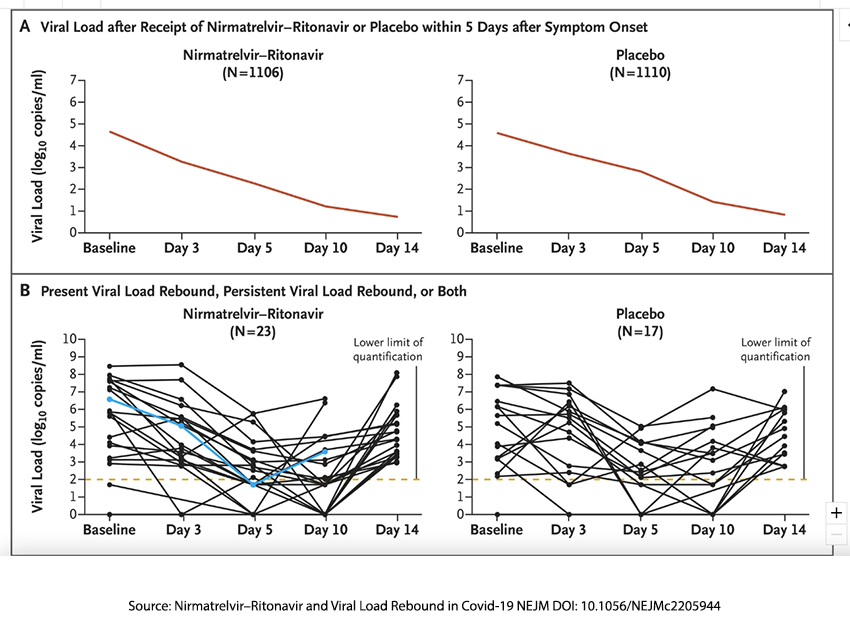
You can see in the top graphic that viral loads for each arm dropped over time, and to be fair, those lines are visually similar enough to suggest that Paxlovid did not accelerate the reduction in viral load compared to the placebo groups’ natural (or acquired through prior infection) immunity. Viral rebound, defined as a “half-log increase on Day 10 or 14, and depicted in the bottom graphic, [2] was seen in 2.3% of Paxlovid patients and 1.7% of placebo patients. As the researchers write,
“Thus, the incidence of viral load rebound was similar in the nirmatrelvir–ritonavir group and the placebo group. The occurrence of viral load rebound was not retrospectively associated with low nirmatrelvir exposure, recurrence of moderate-to-severe symptoms, or development of resistance to nirmatrelvir.”
For us of the general public, we can now add “rebound” to our COVID-19 vocabulary, right there, along with “reinfection.” The data suggest that for a small percentage of us, viral loads, which parenthetically do not necessarily measure viable virus, rebound to a higher level whether it is “effectively treated” or not. As the researcher suggests, the rebound may be a feature of COVID-19’s natural course; more study is needed.
[1] The clinical trial is sponsored by Pfizer; those with a particular mindset may begin throwing stones at the glass house.
[2] The blue line reflects the viral loads of one patient who was hospitalized.
Source: Nirmatrelvir–Ritonavir and Viral Load Rebound in Covid-19 NEJM DOI: 10.1056/NEJMc2205944