
As I've written before, chemistry has a language problem. “Natural” and “organic” are treated as virtues, “synthetic” as something to be avoided—even though these distinctions are often chemically meaningless.
Then there’s a word almost no one uses: semisynthetic. Unlike “natural,” “organic,” or even “synthetic,” this one has real meaning—and this is one of those times. [1]
It has a precise definition: starting with a natural product and modifying it chemically. In at least one case, it made the difference between something that couldn’t be made in useful quantities and a life-saving drug.
There can be no better example than paclitaxel, better known by its original brand name Taxol, a cancer drug used to treat ovarian, breast, lung, pancreatic, and other difficult cancers. It is on the World Health Organization list of essential medicines.
Even today, more than half a century after its discovery, it's not easy to get. Recent shortages and supply disruptions have shown that making enough of the drug remains a challenge.
In fact, in a recent Chemical & Engineering News article, titled Paclitaxel saved my life. New discoveries could boost supply, author Christine Dell'Amore noted that scientists have only just uncovered the final pieces of the biosynthetic pathway that yew trees use to make paclitaxel—underscoring just how extraordinarily complex the molecule is.
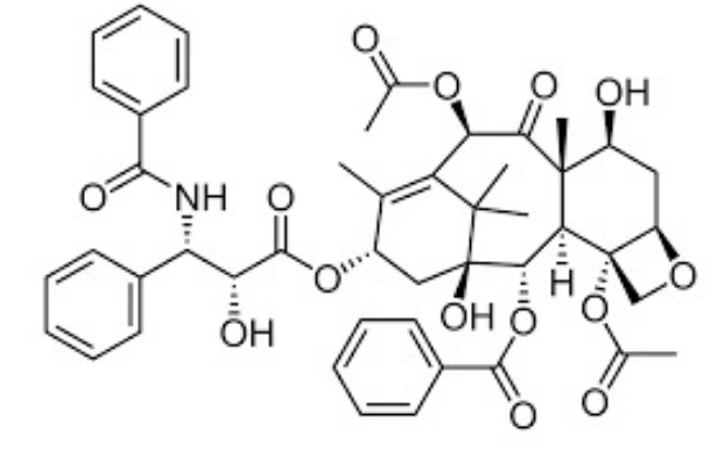
Figure 1. The chemical structure of paclitaxel: An organic chemistry nightmare.
The Taxol Story
Paclitaxel was first isolated from the Pacific yew tree in the 1960s by researchers working with the National Cancer Institute. They weren’t looking for that tree in particular. Instead, they were using a classic drug-discovery approach: collecting thousands of natural samples, grinding them up, and testing crude extracts for biological activity. Many important drugs have been discovered this way.
But the process is anything but simple. Plants contain hundreds of compounds, often in vanishingly small amounts. Crude extracts are tested first. If one shows promise, natural products chemists take over. Their job is to isolate the active compound from a complex mixture and then determine its structure, a process that can take years, especially for molecules as complicated as paclitaxel. It can be crazy-making.[2]
Paclitaxel is one of those molecules. To an organic chemist, its structure means only one thing: trouble. As in the kind of trouble that makes grad students and post-docs consider careers in plumbing or llama farming.
Supply Problems
As the drug moved into clinical trials, two things became clear. First, it worked—astonishingly well in some cancers. Second, there was no way to make enough of it, either by isolating it from its natural source or by making it in the lab.
Nature, as it turned out, wasn’t much help.
Although paclitaxel could be found in different parts of the Pacific yew, the highest concentrations were in the bark—and there wasn’t much of it. The yield from raw plant material to purified drug was about 0.025 percent. In other words, you lose about 99.975 percent of it. To put that into culinary perspective, if baking a cake required 1.5 cups of flour, it would take about 6,000 cups to make the same cake.
Worse, removing the bark kills the tree. A single patient could require several trees, and treating U.S. patients would have meant destroying hundreds of thousands of them. This was never going to work.
The "obvious" alternatives weren’t much better. Finding more of the natural source wasn’t going to happen. Making it from scratch by total synthesis was like climbing the Empire State Building carrying a bulldozer, as I mentioned earlier.
Paclitaxel is so complex that the first total synthesis, completed in 1994, required roughly 40 sequential steps. And no chemical reaction is perfect. Even very good reactions lose material along the way. Multiply those losses over 40 steps, and the overall yield collapses to essentially nothing.
So although multiple research groups succeeded in synthesizing paclitaxel, these heroic efforts had one thing in common:
They produced exactly zero useful supply.
Another solution was needed—one that would combine nature and chemistry.
Enter Robert Holton, a chemist at Florida State University.
Instead of trying to build paclitaxel from scratch, Holton started with a molecule that nature already makes in larger quantities: 10-deacetylbaccatin, found in the needles of the common English yew. Unlike harvesting bark from the Pacific yew, collecting needles doesn’t kill the tree.
From this precursor, Holton devised a short series of chemical steps to convert it into paclitaxel. In doing so, he let nature do most of the hard work and used chemistry only where it was needed (Figure 2).
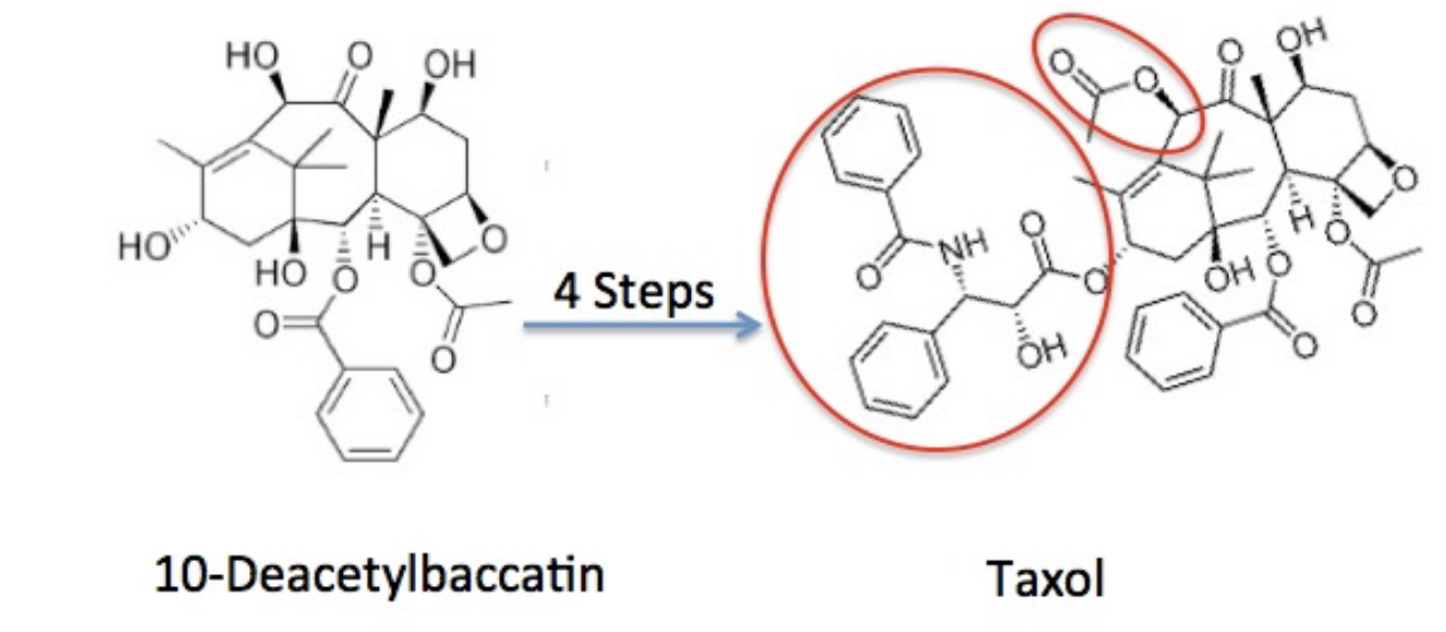
Figure 2. The Holton synthesis. The red ovals highlight the portions of 10-deacetylbaccatin that are chemically modified to produce paclitaxel.
This is semisynthesis—and it solved the problem.
The process was licensed, scaled up, and became the basis for producing paclitaxel in quantities large enough to treat patients worldwide. No mass destruction of trees required.
And even today, paclitaxel is not an easy drug to make. Despite decades of work, recent shortages have shown that supply can still be fragile, and the drug remains expensive to produce. Scientists are only now figuring out how nature assembles the molecule. Turning that knowledge into a practical manufacturing method is another challenge entirely.
In other words, we now understand how nature does it. Reproducing it efficiently is something else. This makes Holton’s solution look even better.
By letting nature do most of the heavy lifting and then finishing the job with a handful of carefully chosen chemical steps, semisynthesis succeeded where brute-force extraction, total synthesis, and even modern biotechnology have fallen short.
“Semisynthetic” is not a marketing term or a proxy for safety; it’s one of the rare labels that actually means something.
In this case, it is the reason the drug exists at all—and why chemistry, not labels, matters.
Notes:
[1] Outside chemistry, the term is often used misleadingly, especially in discussions of opioids, where it describes a drug’s origin but says nothing about potency, safety, or risk. In that context, the distinction between “semisynthetic” and “synthetic” is largely irrelevant.
[2] Natural products mixtures are complex, often containing hundreds of similar compounds. Chemists repeatedly isolate the same naturally abundant molecules while searching for the one responsible for activity, making the process slow and frustrating.



