
A new paper in Analytical Methods reaches a surprisingly simple conclusion: the measured quantity of microplastics may be overestimated due to a simple lab error. More on this below.
I recently jumped all over Netflix for yet another needlessly scary (and predictably one-sided) documentary series. The Plastic Detox followed the Netflix formula:
- Pick a real issue.
- Strip away the nuance.
- Add ominous music
- Quietly omit anything that might reassure the viewer.
The result is a seemingly airtight case against plastics.
Except that when you take a closer look, the airtight case is full of holes.
One of the topics was (of course) microplastics — one of the newer and more insidious scares of the bunch. Is it real? Probably. Has it been overstated? Quite possibly.
It is reasonable to wonder whether there are really that many more tiny bits of plastic all over compared to the past. After all, most plastics in the environment have been in use for decades. So why have they suddenly started “shredding” themselves into ever-tinier, testicle-packing demons?
My reasoning is much the same as that of my colleague, Dr. Joe Schwarcz: when chemicals are unexpectedly detected in the environment, the reason is often improved detection methods, not a sudden influx of something that has been around all along.
As I wrote recently in Yet Another Netflix Improper-ganda Scary Science Show. This Time About Plastic:
“When you improve a microscope lens, the world doesn’t get dirtier; you just start seeing what was already there. Likewise, a better telescope doesn’t create new stars; it reveals ones we couldn’t see before.”
Microplastics are being found everywhere: in oceans, food, air, and, according to some headlines, possibly in your bloodstream. The story is familiar. Tiny plastic particles are accumulating all around (and inside) us, and the numbers keep getting worse.
But according to scientists at the University of Michigan, we may want to pause before taking those numbers at face value. Some of those “microplastics” may not be environmental at all. They may be coming from the lab, the result of faulty measurements caused by contamination from standard nitrile lab gloves — the same kind I used by the tens of thousands as a chemist. In other words, researchers may be detecting their own lab contamination and calling it pollution because material is literally sloughing off their gloves.
The problem hiding in plain sight
Before going any further, it’s worth giving credit where it’s due. The Michigan group did something uncommon: they found an error and publicized it rather than hiding it.
Their microplastics measurements were coming out suspiciously high. Instead of publishing the results and moving on, they stopped to figure out why. The culprit turned out to be sample contamination from their lab gloves. And rather than burying the finding, they published it.
More specifically, the study shows that common laboratory gloves — latex and nitrile — shed microscopic residues during routine handling. These residues, primarily stearates [1] used in glove manufacturing, are problematic because they can be mistaken for microplastics by analytical instruments such as infrared (FTIR) and Raman microscopes, which identify particles based on their chemical signatures.

A Raman microscope. Image: Wikimedia Commons
Raman spectroscopy
Raman spectroscopy is a common analytical method that works by shining a laser on a particle and measuring how the light scatters as it interacts with molecular vibrations. The result is a pattern of peaks — essentially a chemical fingerprint — that can be matched to known materials.
And the resemblance is not superficial. To the instruments, the particles from gloves are indistinguishable from microplastics — not metaphorically, but chemically and physically. They fall in the same size range, appear identical under a microscope, and produce nearly indistinguishable spectral signatures (Figure 1).
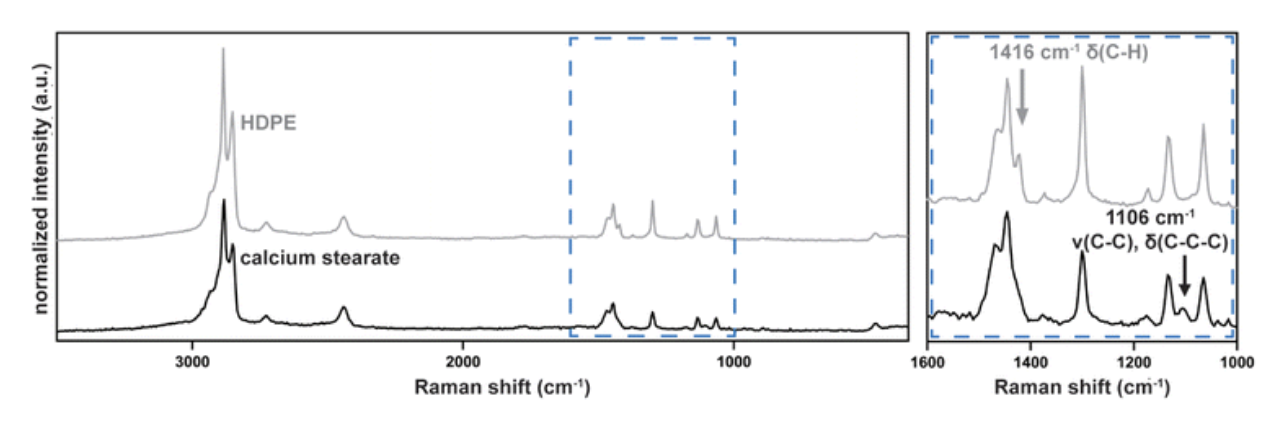
Figure 1. Raman spectra of high-density polyethylene (HDPE) and calcium stearate (a common glove residue). The overall similarity is striking, while the distinguishing features are subtle and confined to specific regions, making misidentification possible without careful analysis. Adapted from Clough et al., Analytical Methods (2024), Fig. 7.
Why this happens
The problem isn’t that the instruments are broken. It’s that they’re doing exactly what they’re designed to do.
Microplastics are typically identified using a similarity score called the Hit Quality Index (HQI). The software compares the chemical “fingerprint” of an unknown particle to a library of known materials and picks the closest match. If the score is high enough, the particle is labeled as plastic.
The catch, as shown in Figure 1, is that stearate residues and common plastics share many of the same dominant chemical features, especially signals from long hydrocarbon chains. As the authors note, the high degree of similarity in these regions can lead to overestimation of microplastics when standard matching methods are used.
In practical terms, the algorithm is doing what any pattern-matching system would do: choosing the closest lookalike. It’s a bit like facial recognition with a blurry camera. If two people share the same major features, the system will confidently pick one — even if it’s the wrong one.
What this means
This doesn’t mean microplastics aren’t real. They are.
But it does mean that some portion of reported microplastic counts — especially the smallest, most concerning particles — may be inflated. And those are precisely the particles most often used to drive alarming claims about human exposure and health effects.
Science works
If anything, this study is a reminder of how science is supposed to work.
The authors didn’t just report surprising results. They questioned them, tracked down the source of error, and published what they found. That’s not a failure — it’s quality control. The more uncomfortable conclusion lies elsewhere:
The field knew contamination was a risk. It just didn’t realize how subtle — and how pervasive — this particular source might be.
Bottom Line
No need.The authors take care of this.
In this work, we illustrated that dry contact with nearly all tested laboratory gloves impart stearate residues to a touched surface that lead to overestimating environmental microplastic quantities[.]
— Clough et al., Analytical Methods, March 26, 2024
NOTES:
[1] Stearates are used in glove manufacturing as lubricants to prevent materials from sticking during production. Small amounts can remain on the surface and transfer during handling.



