The Frank R. Lautenberg Chemical Safety for the 21st Century Act amends the Toxic Substances Control Act (TSCA) and was signed into law June 22, 2016. It created a mandatory requirement for EPA to evaluate existing chemicals with clear and enforceable deadlines, to do so in a transparent fashion, and to do so using risk-based chemical assessments rather than rely on simple epidemiological correlations.
EPA selected the first 10 chemicals to undergo risk evaluation under the amended TSCA and to make those understandable for the public, the American Council on Science and Health is producing risk-based evaluations of each, which will then be compiled into a free downloadable book for consumers and policy makers.
What is Pigment Violet 29?
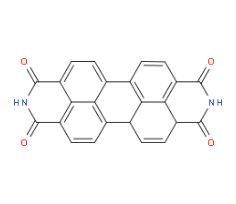
Pigment Violet 29 ((Anthra[2,1,9-def:6,5,10-d'e'f'] diisoquinoline-1,3,8,10(2H,9H)-tetrone or Perylene-3,4:9,10-tetracarboxydiimide)), also known as C.I. Pigment Violet 29, PV29, is a dark red purple (or red violet) solid (powder) and odorless organic compound. It is used to color materials and is an intermediate to create or adjust other perylene pigments and incorporated into paints and coatings used primarily in the automobile industry. It is also incorporated into plastic and rubber products used primarily in automobiles and industrial carpeting. In addition, Pigment Violet 29 is used in merchant ink for commercial printing and in consumer watercolors and acrylic paints.
The chemical structure of Pigment Violet 29 is derived from perylene (a polycyclic aromatic hydrocarbon with the chemical formula C20H12) or acenaphthene, another polycyclic aromatic hydrocarbon, and is shown at the US National Library of Medicine’s Toxnet database (NLM, 2018). Pigment Violet 29 has a molecular formula of C24H10N2O4. Its Chemical Abstracts Service Registration Number (CASRN) is 81-33-4.
Exposure To Pigment Violet 29
Pigment Violet 29 does not readily evaporate and has a low solubility in water and organic solvents. It is not capable of being decomposed by bacteria or other living organisms. It is expected to bind to soil and organic matter and therefore is not expected to migrate through soil to ground water. If it is released into water, it is not expected to evaporate from water into the atmosphere. If Pigment Violet 29 is released into the air, it is expected to be present as particles and is unlikely to break down. Because of this, Pigment Violet 29 is expected to be highly persistent in the environment, but it also has a low potential to accumulate in organisms.
EPA (2017, 2018) has noted that humans are likely to be exposed to Pigment Violet 29 through its manufacturing, production, formulation, handling, and application, through its use in consumer products and/or commercial products, and through a variety of conditions of its use. Workers may be exposed to Pigment Violet 29 through inhalation exposure to particulate matter dust and through skin contact with solids and liquid formulations containing Pigment Violet 29. Consumers may be exposed to Pigment Violet 29 through dermal exposures (through liquid contact) and oral exposures following use of Violet 29-containing products. For the general public, exposures are expected through inhalation of air containing Pigment Violet 29 and through oral exposures that may result from the conditions of use of Pigment Violet 29.
Although the manufacturing, processing, distribution, use and disposal of Pigment Violet 29 can result in release, no monitoring data have been identified for Pigment Violet 29 in outdoor or indoor house air, drinking water, surface water, soil, sediment, organisms, or food in the US. However, a domestic manufacturer of C.I. Pigment Violet 29 reported a workplace air concentration of 0.5 mg/m3 over a 12-hour shift (EPA, 2018). EPA noted that it is not clear if the report was for C.I. Pigment Violet 29 or for total dust. Thus, the actual air concentration of C.I. Pigment Violet 29 may be lower than 0.5 mg/m3.
Health Effects Of Pigment Violet 29
C.I. Pigment Violet 29 is expected to be poorly absorbed by all routes (oral, dermal, and inhalation) due to its large and bulky structure (EPA, 2018). No hazard has been identified in humans following exposure to Pigment Violet 29 by any route. However, EPA (2018) has noted that an initial screening conducted by other regulatory agencies indicates that the hazards to human and environmental receptors following exposure to Pigment Violet 29 are expected to be low.
Based on the limited data on the toxicity Pigment Violet 29 in experimental animals (ECHA, 2017), brief high levels of Pigment Violet 29 were not toxic following inhalation, oral or dermal exposure. Short-term oral exposure to high levels of Pigment Violet 29 also did not result in adverse health effects. In addition, oral high levels of Pigment Violet 29 were not toxic to reproduction or development of the offspring. There were no lifetime studies in animals.
Although testing for carcinogenicity of Pigment Violet 29 has not been conducted, EPA (2018) concluded that available data on Pigment Violet 29 suggests that it is unlikely to be a carcinogen. Furthermore, because key data are missing on the toxicity of Pigment Violet 29, EPA (2018) will be conducting a risk evaluation for Pigment Violet 29.
Pigment Violet 29 Safe Levels
Both the federal and state government develop regulations and recommendations to protect public health, often expressed as a safe or virtually safe level, that is, a level of a substance in air, water, soil, or food that is not expected to cause any adverse health effect, even in people who are sensitive to the chemical’s effects.
These safe levels are usually based on information from experiments with animals (usually rodents) at much higher levels of the chemicals than humans would typically encounter. The higher animal exposures are used to see what adverse health effects could occur. Scientists then conjecture what the adverse effects could possibly be in humans at a lower level of exposure. Scientists can then estimate the level that will likely protect humans.
Sometimes these safe levels differ among federal and state organizations because they used different assumptions for human exposure, different animal studies, or employ methods that differ slightly. Other times, these recommendations differ because new science develops that suggests different levels are toxic or safe. Recommendations and regulations are also updated periodically as more information becomes available.
So far, due to no hazard identified in humans, and no lifetime animal studies, no organization has derived a safe or virtually safe level for Pigment Violet 29. Furthermore, internal doses of Pigment Violet 29 have not been estimated that correspond to administered doses in experimental animals.
The ratio of an internal or external dose to the human exposure is referred to as a Margin of Exposure (MOE). This MOE can also be used as a threshold to determine the presence or absence of risk. To assess risks, the MOE is interpreted as a risk of concern if the MOE is less than the benchmark value (usually 100). On the other hand, the MOE indicates negligible concerns for adverse human health effects if the MOE exceeds this benchmark value. Typically, the larger the MOE, the more unlikely it is that an adverse effect would occur.
If available, an internal or external dose of Pigment Violet 29 could be divided by actual or anticipated human exposures to determine a ratio, referred to as a margin of exposure approach.
Why Is EPA Looking At Pigment Violet 29 Under The Lautenberg Chemical Safety Act?
EPA scientists are currently looking at the likely routes of exposure to Pigment Violet 29 in the environment and will be developing exposure scenarios, or pathways, of how the public comes into contact with Pigment Violet 29. These exposure pathways will then be studied by EPA by comparing the amount of Pigment Violet 29 exposure in the pathway to its safe or virtually safe level, or to the internal dose associated with decreased fetal body weights (as noted above).
If human exposure in the pathway is at or below this safe or virtually safe level, then Pigment Violet 29 exposure from the pathway is not considered to be a human health concern. If exposure is above this safe or virtually safe level, then the pathway might be considered as a possible health concern; small excesses of the safe or virtually safe dose are seldom cause for concern since these safety levels are developed from conservative assumptions, including the use of safety factors that tend to exaggerate risk and exposure pathways that tend to exaggerate exposure.
several pathways may be added together to suggest a health concern. In either event, regulations might be developed to lessen the exposure of Pigment Violet 29 from this pathway(s). See also EPA (2018) for specific questions related to the assessment of Pigment Violet 29 under the new Lautenberg Chemical Safety Act (LCSA).
Controversy Over Pigment Violet 29
Although EPA has indicated that human and environmental health hazards of Pigment Violet 29 are low, health impacts of Pigment Violet 29 have not generally been assessed. Therefore, EPA (2018) has indicated that a hazard evaluation will be conducted for Pigment Violet 29, where it plans to judge the appropriateness or relevance of any hazard within the context of a specific exposure scenario.
However, EPA expects C.I. Pigment Violet 29 exposures to be low. Because of the apparent low exposures to environmental and human health of Pigment Violet 29, EPA (2018) does not plan to further analyze numerous environmental and exposure pathways for any population. Exclusion of this broad group of exposure scenarios from further analysis may prove to be controversial.
Lacking any evidence of hazard in humans or lifetime animal models, the American Council on Science and Health has recommended to the Environmental Protection Agency that Pigment Violet 29 not be banned.
More analyses in our series on the Lautenberg Chemical Safety Act compounds:
ACSH Explains: What's The Story On N-Methylpyrrolidone?
ACSH Explains: What's The Story On Cyclic Aliphatic Bromides Cluster (HBCD)?
ACSH Explains: What's The Story On Carbon Tetrachloride?
ACSH Explains: What's The Story On Bromopropane?
ACSH Explains: What's The Story On Dioxane?
ACSH Explains: What's The Story On Trichloroethylene (TCE)?
ACSH Explains: What's The Story On Methylene Chloride (DCM)?
ACSH Explains: What's The Story On Asbestos?
REFERENCES:
ECHA (European Chemicals Agency). (2017). ECHA registration dossier: Perylene-3,4:9,10-tetracarboxydiimide. CAS number: 81-33-4. (EC number: 201-379-7). Helsinki, Finland. Available at: https://echa.europa.eu/registration-dossier/-/registered-dossier/10330
EPA (U.S. Environmental Protection Agency). (2017). Scope of the risk evaluation for Pigment Violet 29 (Anthra[2,1,9-def:6,5,10-d'e'f']diisoquinoline-1,3,8,10(2H,9H)-tetrone): CASRN: 81-33-4 [EPA Report]. (740-R1-7011). U.S. EPA, Office of Chemical Safety and Pollution Prevention, Office of Pollution Prevention and Toxics. Available at: https://www.epa.gov/sites/production/files/2017-06/documents/pv29_scope_06-22-17.pdf
EPA (U.S. Environmental Protection Agency). 2018. Problem Formulation of the Risk Evaluation for C.I. Pigment Violet 29 (Anthra[2,1,9-def:6,5,10-d'e'f']diisoquinoline- 1,3,8,10(2H,9H)-tetrone) Office of Chemical Safety and Pollution Prevention. EPA Document# 740-R1-7021. Available at: https://www.epa.gov/sites/production/files/2018-06/documents/pv29_proble...
NLM (U.S. National Library of Medicine). 2018. ChemIDplus: Pigment Violet 29. Toxnet database. Found at: https://chem.nlm.nih.gov/chemidplus/rn/81-33-4
Data provided by
Michael Dourson, PhD, DABT, FATS, FSRA
Bernard Gadagbui, MS, PhD, DABT, ERT
Bethany Hansen, MA
all of Toxicology Excellence for Risk Assessment (TERA)