
Few biological trends generate as much public anxiety as the perception that children are growing up too fast. Over the past century, the average age at which puberty begins has declined dramatically. Improvements in hygiene and nutrition are often cited to explain that decline. In recent decades, that pubertal shift has fueled concern that synthetic chemicals in our environment are disrupting children’s endocrine systems. The narrative is compelling: endocrine-disrupting chemicals (EDCs) interfere with hormonal signaling, puberty is hormonally driven, therefore chemicals must be accelerating development.
But biology rarely conforms to single-cause explanations.
Puberty is not triggered by a lone hormone or a single environmental exposure. It is the product of a tightly regulated neuroendocrine network integrating genetics, metabolic status, stress signaling, and environmental inputs. A large, longitudinal analysis from the Danish National Birth Cohort offers an opportunity to move beyond speculation and examine how these factors interact in real populations. Spoiler alert: as with most complex biological phenomena, the answer is less about one villain and more about competing signals within a delicately balanced system.
Understanding what is shifting the timing of puberty requires understanding how the reproductive axis works in the first place.
Menarche: What Is “Early,” Really?
Precocious or early puberty is defined by the “premature” development of sexual characteristics and accelerated growth. Most often, early puberty is idiopathic, attributed to unknown causes, although there are rare instances where genetic conditions or abnormalities in the nervous system are found. Definitions matter because what we label as “early” depends partly on historical and population norms.
Menarche, the onset of the female reproductive capacity, is a component of puberty and is often identified with those secondary sexual changes as the marker of puberty’s onset. As it turns out, our contemporary use of the term “premature” fails to account for human history. A study of skeletal remains suggests a far earlier onset between 7 and 13 years for Paleolithic women settling during medieval times, to reports approximating 14 years of age. With the industrial revolution and the vast migration to cities, followed by malnutrition and deteriorating living conditions, menarche rose to 15 to 16 years of age. It seems that the improvements in hygiene and nutrition haven’t reduced the age of menarche as much as they have brought it back into an earlier alignment. That said, today it seems as though the downward trend has slowed.
With that historical context in mind, the next question becomes mechanistic: how does the body decide when to begin?
How Puberty Works: The Brain–Endocrine–Reproductive Axis
The neuroendocrine changes that bring about puberty are complex—but understanding them clarifies how genetics, metabolism, stress, and chemicals might influence timing. Here is the gist, but for those TL;DR amongst us who favor a simple diagram, you can find it at the end of this section.
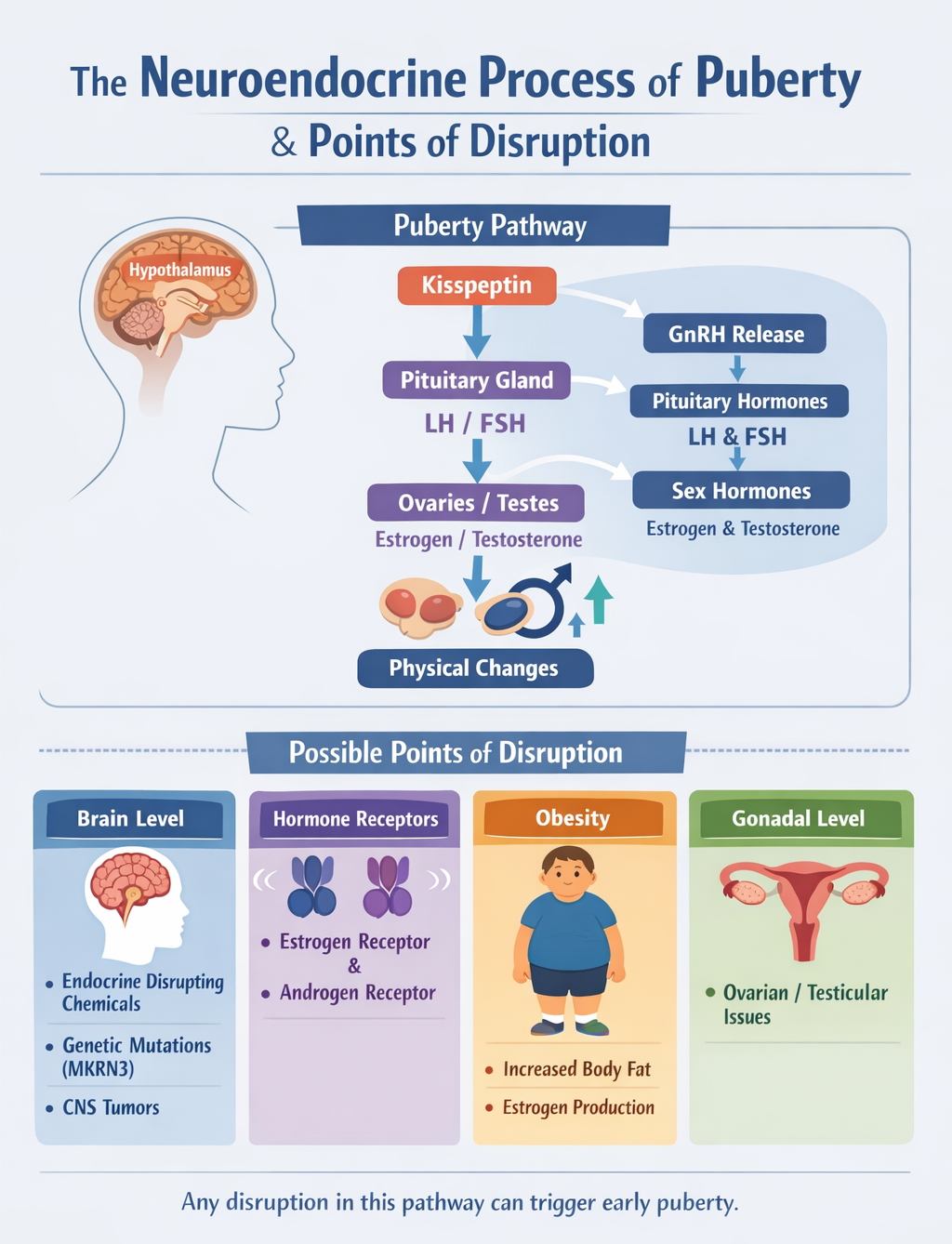
Puberty is initiated and regulated by a cascade of hormonal signaling, the reproductive axis, that begins in the brain and extends through the endocrine system to the reproductive organs.
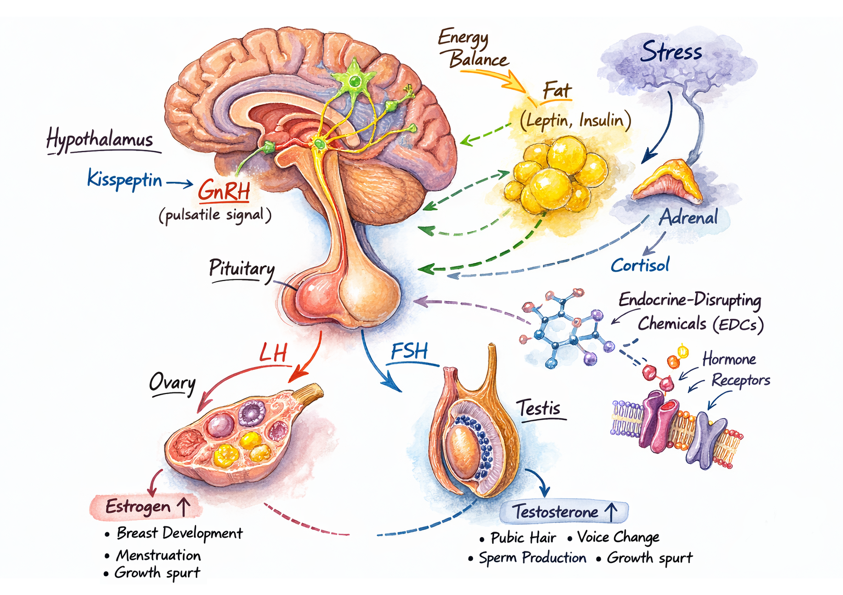 Puberty begins in a small brain structure called the hypothalamus, which rhythmically secretes gonadotropin-releasing hormone (GnRH), acting as the metronome of the reproductive axis. Kisspeptin, a neuropeptide, stimulates the GnRH neurons, initiating puberty.
Puberty begins in a small brain structure called the hypothalamus, which rhythmically secretes gonadotropin-releasing hormone (GnRH), acting as the metronome of the reproductive axis. Kisspeptin, a neuropeptide, stimulates the GnRH neurons, initiating puberty.- GnRH stimulates the pituitary gland to release Luteinizing hormone (LH) and Follicle-stimulating hormone (FSH), two hormones that enter the bloodstream to stimulate the ovaries and testicles to produce estrogens and testosterone.
- These hormones drive breast development, testicular enlargement, pubic hair growth, menstruation, and growth spurts. Eventually, estrogen causes closure of the bones’ growth plates, stopping growth.
Genetic disruptions can either remove inhibition, leading to early puberty, or prevent activation, resulting in delayed or absent puberty. Some endocrine-disrupting chemicals (EDCs) in the environment can activate kisspeptin or GnRH receptors, thereby prematurely activating the reproductive axis. Those same EDCs can also bind to estrogen and androgen receptors, mimicking or blocking natural hormones and disrupting hormonal action rather than production. While laboratory studies demonstrate these mechanisms under controlled conditions, real-world human exposures are typically far lower and more variable, complicating direct translation.
Fat cells are a potential source of estrogen, converting androgens into estrogens. Obesity influences the onset of puberty by acting as an “energy” signal to the brain, triggering the process. Breast tissue, highly sensitive to estrogen, may develop earlier in those with obesity, creating an asynchronous puberty in which secondary changes occur far sooner than ovarian maturation.
Puberty is fundamentally a brain-driven neuroendocrine cascade. Any factor that prematurely activates GnRH, removes inhibitory signals, mimics hormones, alters receptor activity, or shifts metabolic energy cues can potentially alter its timing. Early puberty likely reflects disruption at multiple levels of this tightly regulated system rather than a single point of failure.
The Danish National Birth Cohort: Design and Scope
The Danish National Birth Cohort (DNBC) is a nationwide cohort study that recruited pregnant women in Denmark between 1996 and 2002, enrolling about 30% of all pregnant women. Participants provided detailed health and lifestyle information at enrollment, during pregnancy, and in early childhood. The DNBC Puberty Cohort includes children born to DNBC participants. Of 56,641 eligible children, 22,439 were sampled and invited to participate; 15,819 (8,123 girls and 7,696 boys) responded, yielding a participation rate of 70%. From 2012 to 2021, at ages 11 to 18 years, they provided information on several pubertal milestones every six months.
The study investigated a range of potential causes of early puberty, including genetic factors, maternal lifestyle and dietary factors, maternal diseases and medication use that may result in prenatal exposure to EDCs, birth-related and postpartum events, children’s health and anthropometrics, and prenatal and early-life exposure to psychosocial factors.
Genetics, Weight, Stress—and Chemicals?
- Girls began puberty at an average age of 10.5 years and reached menarche at 13.0 years. Girls reached menarche an average of 3.6 months earlier than their mothers, indicating a significant decline in age at menarche across one generation
- Boys began puberty at 11.1 years, experienced voice break at 13.1 years, and had their first ejaculation at 13.4 years.
Several thematic associations were evident in the researcher’s findings.
- Earlier maternal age at menarche predicted earlier puberty in both daughters and sons, supporting a shared genetic influence.
- Maternal smoking and obesity were also associated with earlier puberty. Maternal medications had little effect.
- Childhood obesity was associated with earlier puberty.
- Maternal and childhood stress were associated with modestly earlier puberty.
- PFAS exposure showed mixed and variable associations, none of which reached statistical significance.
The association between stress and earlier puberty is biologically plausible. When we experience stress, we release cortisol. Cortisol also communicates with the reproductive axis, altering the pulsatile release of GnRH and downstream reproductive hormones, all of which are key drivers of puberty. Stress can also shift metabolic signals such as leptin and insulin, which, like obesity, signal energy readiness for reproduction, further influencing pubertal timing. The direction of effect likely depends on timing, duration, developmental stage, and metabolic context.
Taken together, the pattern suggests stronger and more consistent signals from inherited timing and metabolic factors than from measured chemical exposures in this cohort.
What Matters Most and What Matters Less
Across categories, three factors emerge with the most consistent associations: genetic predisposition (as reflected by maternal age at menarche), maternal smoking during pregnancy, and childhood obesity. Of these, only genetics is immutable. The strongest and most modifiable factor is body weight.
By contrast, exposure to PFAS, emblematic endocrine disruptors, showed mixed and statistically nonsignificant associations in this cohort. That does not mean environmental chemicals play no role; rather, in this cohort, their measurable associations were weaker and less consistent than those observed for genetic and metabolic factors.
When it comes to pubertal timing, the dominant signal may not be synthetic chemicals that mimic hormones but rather energy-abundance signaling that indicates reproductive readiness.
Puberty as a Systems Problem
The evidence surrounding earlier puberty raises a central question: what, exactly, is driving the shift? Framing puberty as a systems problem offers a clearer answer.
Puberty is not a switch flipped by a single exposure. It is an orchestrated neuroendocrine cascade integrating genetic programming, metabolic status, stress signaling, and environmental inputs. Because the process begins in the hypothalamus and depends on pulsatile hormonal signaling, it is exquisitely sensitive to multiple converging signals.
The Danish cohort data suggest that while endocrine-disrupting chemicals remain biologically plausible contributors, they are not the primary drivers detectable at the population level. Instead, inherited timing patterns, maternal smoking, psychosocial stress, and most consistently, childhood obesity emerge as more influential factors.
The modern environment does influence puberty. But the most powerful environmental signal may not be trace chemicals in plastics; it may be sustained caloric abundance and altered metabolic signaling in developing children.
Biology, once again, resists simple narratives. Puberty is not being “hijacked” by a single disruptor; it responds to the totality of the signals we provide. And in that equation, energy balance appears to speak louder than endocrine mimics.
Source: Ten-year findings from the Puberty Cohort: a sub-cohort within the Danish National Birth Cohort International Journal of Epidemiology, DOI:10.1093/ije/dyaf209



