
Coal Tar’s Magic Bullet
Methylene blue was first synthesized in the German dye industry, when many synthetic coal-tar dyes were being developed. It had no medical purpose; it was simply a vivid blue textile dye. Its first biological value came when scientists realized that it could selectively stain. Within a few years, it was one of the first vital stains, helping researchers visualize nerve cells and other tissues.
That same chemical versatility caught the attention of Paul Ehrlich, a pioneer of early antimicrobial therapy. He used methylene blue to treat malaria, not because it stained the parasite, but because it interfered with its metabolism—an early example of his “magic bullet” concept. [1] Beyond its diagnostic role in tracing physiological pathways, methylene blue remains an FDA-approved treatment for methemoglobinemia, a condition in which hemoglobin cannot effectively carry oxygen.
Today, it is being promoted by a host of influencers, from biohacker Bryan Johnson, who, as part of his operation Blueprint, began using methylene blue, which ironically turned his urine blue, to social commentator Joe Rogan and, apparently, even Secretary Kennedy. [2] However, to understand the renewed interest in methylene blue, we need to consider, as Joe Rogan points out, its “profound effects on your mitochondria.”
The Machinery of Cellular Power
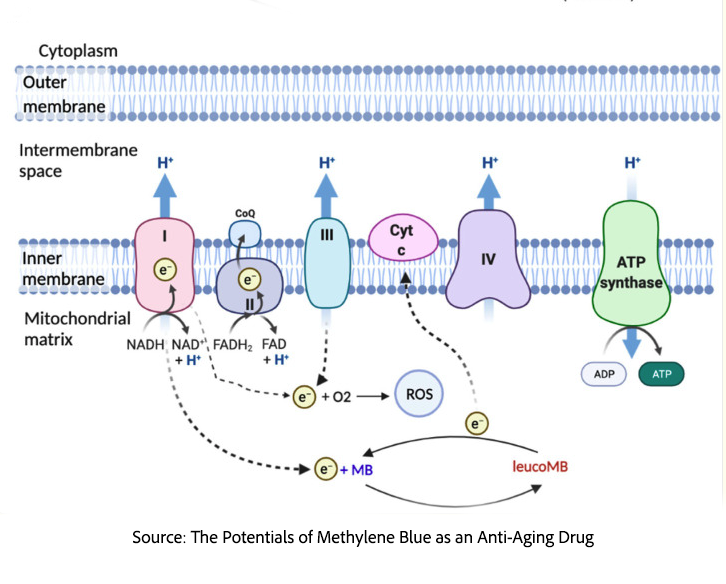
Mitochondria are thought to be free-living bacteria that our primitive cellular ancestors formed an endosymbiotic partnership with several billion years ago. In exchange for living safely within our bodies, mitochondria serve as the primary sites of cellular energy production, converting the chemical energy stored in the nutrients we consume into ATP, which powers our cellular activities.
The conversion is carried out by the mitochondrial electron transport chain, a carefully engineered energy-processing production line, pictured above. Within the mitochondria, electrons function as packets of chemical energy, and their movement is governed by differences in the tendency of a molecule to accept or donate electrons, its redox potential.
The electrons arrive carried by NADH and FADH2, molecules that are not particularly inclined to hold onto them. These electrons pass through a series of protein complexes embedded in the inner mitochondrial membrane, ultimately reacting with oxygen, a highly electron-hungry molecule. As they move “downhill,” they release small amounts of energy at each step.
Rather than letting that energy dissipate as heat, the cell captures it in an intermediate storage by pumping protons (hydrogen ions) from the mitochondrial matrix into the space between the membranes. This creates both a concentration gradient, with more protons outside than inside, and an electrical difference called the proton motive force. Like water building up behind a dam, the energy originally carried by electrons is now stored as a spatial and electrical gradient.
Enter the turbine in our energy dam, ATP synthase (Complex V). This is a moment of biological magic. Protons want to flow backward to reduce their concentration and electrical gradients. As they pass through a portion of the enzyme, they cause a ring structure within the enzyme to rotate. This mechanical force drives ADP and inorganic phosphate to bind and be squeezed together to form ATP, which is then released, as the cell’s primary energy currency. The system transforms electrochemical energy into mechanical rotation and then into chemical bond energy.
The process, however, is not perfectly controlled. When electrons accumulate or stall, particularly at Complex I, they can leak out and react prematurely with oxygen, producing the chemically damaging reactive oxygen species (ROS). Methylene blue introduces an auxiliary “scavenger” pathway because it readily switches between accepting and donating electrons. It can accept electrons from NADH and deliver them directly to cytochrome c, further down the chain, thereby keeping electrons moving and reducing the risk of leakage and ROS formation. In doing so, methylene blue helps preserve both electron flow and proton gradient buildup, stabilizing ATP production even when parts of the native system are impaired.
This biochemical role is what makes methylene blue particularly intriguing in diseases linked to mitochondrial dysfunction.
Promise vs. Proof
Methylene blue is biologically interesting because it targets a central vulnerability in brain aging, namely mitochondrial dysfunction that accompanies aging and contributes to neuronal loss and diseases such as Alzheimer’s and Parkinson’s.
Methylene blue stands out because it readily crosses the blood-brain barrier and accumulates in brain tissue; it has a strong affinity for mitochondria and, by bypassing parts of the electron transport chain, can restore mitochondrial membrane potential and energy function.
Early in Alzheimer’s disease, mitochondria shrink, producing less energy and more oxidative stress. The hallmark lesions of amyloid plaques and tau tangles increase in the face of mitochondrial dysfunction and, in turn, may further mitochondrial damage. That being said, it is unclear whether either is causative, although methylene blue has been shown to clear amyloid and tau aggregates, suggesting it might break the presumptive vicious cycle between protein aggregation and mitochondrial failure.
Unfortunately, despite a plausible physiologic pathway, the clinical evidence is mixed. While early studies suggested reductions in cognitive decline, phase III studies were inconclusive – in short, the evidence is promising but not definitive.
Similarly, in Parkinson’s disease, characterized by the loss of dopaminergic neurons and aggregates called Lewy bodies, there is a strong association with mitochondrial dysfunction and oxidative stress. However, human evidence is lacking, so the role of methylene blue in Parkinson’s remains uncertain.
As in neurodegenerative disease, aging skin experiences oxidative stress and mitochondrial dysfunction, making methylene blue a plausible treatment. The skin is the largest organ and the one that contains all the other parts. Methylene blue absorbs UV rays and supports wound healing by stimulating fibroblasts, elastin, and collagen – components of our reparative process. Again, what is biologically plausible and seen in the laboratory has little human evidence of beneficial outcomes. Human studies are few and small and report skin improvement in qualitative, not quantitative, ways. Bottom line, there are no large, well-designed clinical trials demonstrating methylene blue’s “anti-aging effects.”
A Grifter’s Dream
Methylene blue sits at a “Goldilocks” intersection of biological plausibility, pre-clinical laboratory evidence, and a few clinical studies designed more for marketing than scientific evidence. And, like Goldilocks, the story begins with "once upon a time" and ends with "happily ever after," without being grounded in an adult reality.
Methylene blue comes wrapped in the language of mitochondria, oxidative stress, electron transport, neuroprotection, and longevity—terms that let promoters cosplay precision while selling speculation. The evidence base is perfect for hype: intriguing mechanisms, positive findings in cells and animals, scattered human signals, and just enough ambiguity to let every caveat be recast as suppression, closed-mindedness, or the tragic slowness of mainstream medicine.
Because the compound has legitimate medical uses, the sales pitch advances unsubstantiated claims. And because consumers cannot easily distinguish between “biologically interesting,” “preliminarily promising,” and “clinically proven,” methylene blue becomes an ideal vessel for influencer medicine: vivid, contrarian, just complicated enough to intimidate skepticism, and always one podcast episode away from immortality.
[1] Paul Ehrlich’s “magic bullet” was his idea of designing a chemical that would selectively kill a disease-causing organism without harming the host. The most famous example was Salvarsan (arsphenamine), used to treat syphilis, a major and often deadly disease at the time.
[2] Oddly, methylene blue is part of the same family of synthetic dyes that Secretary Kennedy has been adamant about getting out of our food supply.
Sources: The Potentials of Methylene Blue as an Anti-Aging Drug. Cell DOI: 10.3390/cells10123379
Methylene Blue StatPearls



