Why cholesterol measurement is changing
Heart disease remains the leading cause of death in the US and with a projected costs of $1 trillion in 2035, a major driver of healthcare costs. Our treatment, based on a model of dysfunctional lipid metabolism being the proximate cause of cardiovascular disease has focused on LDL, the “bad cholesterol.” But newer American College of Cardiology and American Heart Association guidelines, built around updated risk calculation, encourage clinicians to treat earlier, measure more precisely, and intervene more aggressively when warranted.
As Ciaran Kohli-Lynch, an epidemiologist at Northwestern and lead author of the new study, put it:
“This means it is increasingly important to accurately identify who would benefit most from intensive treatment.”
Before diving into the study, let’s briefly discuss the lipoproteins involved in cardiovascular disease.
The metabolic “bad boys” of cardiovascular disease
Apolipoprotein B (ApoB) is present on the lipoproteins we have identified as causal factors in atherosclerotic cardiovascular disease. It is found on low-density lipoprotein cholesterol (LDL), the poster child of bad cholesterol, and is also present on the less concerning posse of very LDL-C, intermediate-density lipoprotein cholesterol, and lipoprotein A. Because these particles can contribute to plaque buildup in arteries, lowering atherogenic lipids remains a cornerstone of primary prevention.
But LDL is not the only way to assess risk. LDL-C estimates how much cholesterol is carried in LDL particles, while non-HDL cholesterol captures a broader pool of atherogenic particles. ApoB adds another layer: because each harmful particle carries one ApoB molecule, ApoB serves as a rough count of the number of particles. In that sense, LDL is the simplest measure, while non-HDL and especially ApoB may offer a more complete picture of risk.
As Kohli-Lynch argues, ApoB may identify risk better than LDL because it reflects the total number of harmful particles circulating in the blood.
Because all ApoB-containing lipoproteins can enter arterial walls and initiate the formation of atherosclerotic plaque, ApoB is often considered a more precise marker of cardiovascular risk, especially when triglycerides are high or LDL appears “normal.”
ApoB is not part of the standard lipid panel most physicians order. So the study asked a practical question: if ApoB is a more precise marker of cardiovascular risk, is it worth the extra testing cost to use it instead of LDL to guide treatment?
That question led the researchers away from the clinic and into a computer model designed to estimate how different testing strategies might play out across an entire population.
Modeling Treatment
Researchers used a computer model representing 250,000 US adults to estimate the lifetime risk of heart disease, the cost of care, and quality of life in a cohort without prior cardiovascular disease, tracking individuals over time to mimic longitudinal prevention and treatment. The simulated population was built from NHANES and other health surveys, including key cardiovascular risk factors, i.e., cholesterol levels, blood pressure, diabetes, and smoking providing a population targeted for preventative care.
All patients began with guideline-based statin therapy, and the model then compared three ways of deciding whether to intensify treatment:
- Traditional LDL cholesterol targets of <100 mg/dL
- Non-HDL cholesterol targeted at <118 mg/dL
- ApoB (a marker of particle number) targeted at <78.7 mg/dL
Each approach escalates therapy if targets aren’t met, reflecting real-world stepwise care that mirrors clinical reality, where patients respond differently and treatment effects are not uniform. The newer markers (non-HDL and apoB) tend to drive more aggressive treatment.
The simulated population reflects a typical real-world primary care group—older adults at meaningful cardiovascular risk, diverse in background, and eligible for statins. By evaluating health benefits, cost-effectiveness, and value for money, along with the downside risks associated with side effects and costs, the model captures the trade-offs clinicians weigh when intensifying therapy.
With that setup in place, the study could ask the question that matters most to patients and payers alike: what do these different strategies actually buy?
More prevention, better outcomes
“We found that ApoB testing to intensify cholesterol-lowering medication would prevent more heart attacks and strokes than current practice, and that these health benefits were achieved at a cost that represents good value for U.S. healthcare payers,” - Ciaran Kohli-Lynch
Using standard LDL-based care as the baseline, the non-HDL strategy prevented an additional 617 cardiovascular events, added 965 quality-adjusted life years, and reduced overall costs by about $2.1 million. It also increased the share of patients receiving more intensive treatment from 50% to 57%, suggesting that it identified more people whose risk warranted stronger therapy without sacrificing efficiency at the population level.
The ApoB-guided strategy was more aggressive still. It intensified therapy for 64% of patients, prevented 1,018 cardiovascular events, and added 1,324 quality-adjusted life years, but it also increased costs by about $40 million.
A Cost trade-off
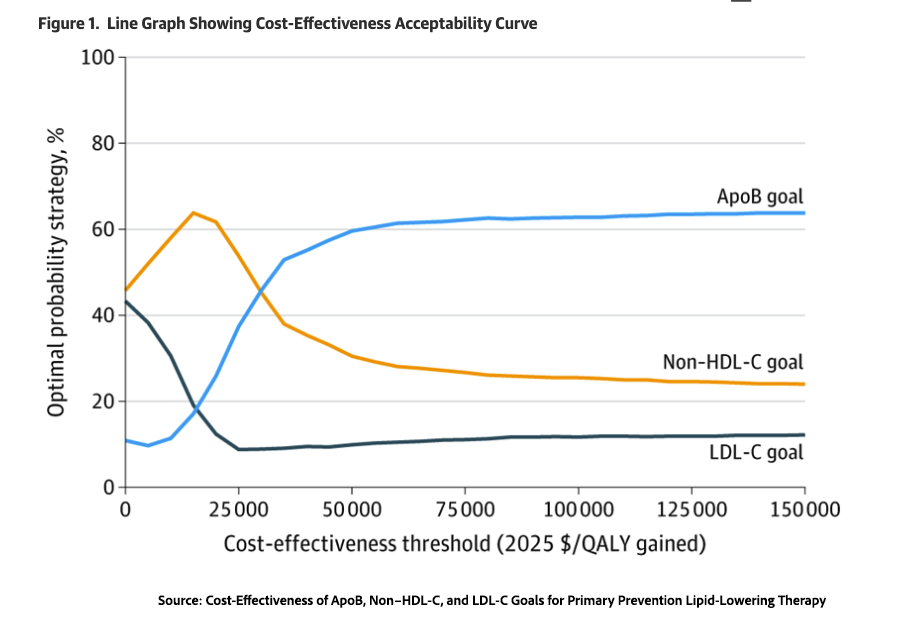
Medical economics uses several methods to quantify cost in these models. One key measure is the Incremental Cost-Effectiveness Ratio (ICER), which answers the question of how much extra health we get for the money spent when switching to a new treatment. Under the non-HDL guidance, the ICER showed annual savings for men and a $107 cost for women. By contrast, the more aggressive ApoB treatment had annual ICER values of “$35 900 per QALY for women and $26 600 per QALY for men.”
Another way to frame the same issue is through a willingness-to-pay threshold: how much society is willing to spend to gain one quality-adjusted life year. In the United States, that threshold is often set at $100,000 to $120,000 per QALY. By that standard, the study suggests that both alternative strategies were economically reasonable, even though ApoB required more upfront spending.
The researchers also highlight an irony: the added costs of ApoB guidance do not come mainly from the test itself. They stem from the longer lives it helps produce, along with the healthcare costs that accumulate over those additional years. In other words, preventing disease can increase total spending simply because people live long enough to need more care later.
For advocates of precision medicine, the study underscores an important trade-off. As treatment becomes more biologically refined, prevention improves: fewer cardiovascular events occur, and patients gain more healthy years. But the economics also shift. There may be less spending on acute heart disease and more spending on prevention and care delivered across longer lives. Best care and cost are, dare I say, complicated and nuanced?
The best strategy, then, is not simply the cheapest one or the most precise one, but the one whose benefits justify its broader costs.
[1] Quality-adjusted life years, or QALYs, are a standard way of combining longer life and better health into a single measure.
Source: Cost-Effectiveness of ApoB, Non–HDL-C, and LDL-C Goals for Primary Prevention Lipid-Lowering Therapy JAMA DOI: 10.1001/jama.2026.2986



