At first glance, drug law enforcement and drug resistance seem to have nothing whatsoever in common. One involves microbes evolving in hospitals and laboratories; the other involves law enforcement trying to disrupt the illicit drug trade. But both systems are governed by the same fundamental principle: evolution under selective pressure.
Anyone who has worked in antiviral or antibacterial research knows the pattern well. When drugs kill susceptible microbes, rare variants carrying resistance mutations survive and multiply. Over time, those resistant strains can dominate the population.
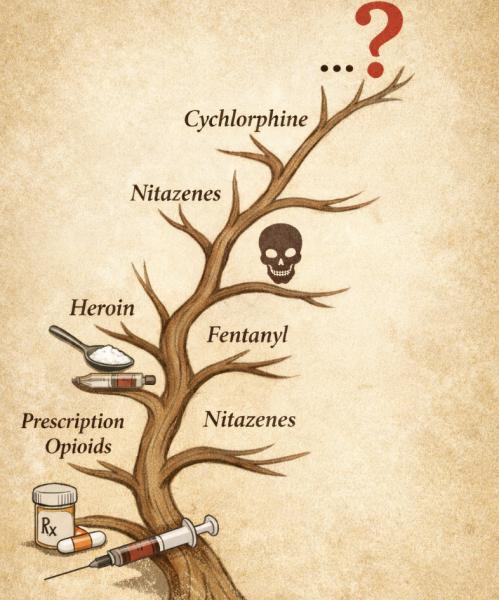
Something remarkably similar happens in the illicit drug market. When authorities eliminate one drug or restrict its supply, the market adapts by producing alternatives that evade those constraints—much as we are now seeing with fentanyl.
The result is another new, potentially more dangerous opioid now appearing in forensic laboratories: cychlorphine.
Authorities in several countries have recently detected cychlorphine in the illicit drug supply, with reports in the United States emerging more recently. Forensic monitoring systems first detected it in parts of Europe around 2024. By 2025, it appeared in drug-checking programs in Toronto and in seized drug samples in southwest Ohio, and since then, authorities have reported detections or related overdose deaths in places such as eastern Tennessee, central Kentucky, and the Chicago area.
Chemically, cychlorphine belongs to an entirely different family from fentanyl and the nitazenes. Its synthesis can rely on industrial intermediates that also have many legitimate uses, making it far harder for regulators to track production by monitoring a small set of precursor chemicals. In medicinal chemistry terms, cychlorphine represents a shift to a different molecular scaffold—one capable of producing powerful opioid effects without relying on the same ingredients used to manufacture fentanyl.
If the pattern holds—as it has repeatedly over the past half-century—cychlorphine will not be the last new opioid to emerge from the cat-and-mouse game between drug prohibition and illicit innovation.
Like many synthetic opioids before it—including the nitazenes that appeared in the illicit market a few years ago—cychlorphine rarely shows up as a labeled product. Instead, it often appears mixed into counterfeit pills or polysubstance mixtures, another sign that traffickers are already experimenting with the next generation of synthetic drugs.
At first glance, cychlorphine might appear to be just another version of fentanyl. But that conclusion is wrong for two reasons.
First, fentanyl production eventually became easier for regulators to track because its synthesis converges on a small number of well-known intermediates that regulators learned to monitor. Even more potent fentanyl analogs share much of fentanyl’s core structure, so they often require some of the same precursor chemicals.
With cychlorphine, however, all bets are off. Although cychlorphine and fentanyl share some recognizable structural elements, much of the molecule is different. As a result, the list of precursor chemicals used to track fentanyl production is likely to be of little help in identifying cychlorphine manufacturing.
Worse still, a structurally different opioid can still bind to the same brain receptor as morphine—only much more tightly. Even the most experienced organic chemist could look at the structures of morphine and cychlorphine and see essentially no resemblance.
This makes predicting the next novel opioid extraordinarily difficult.
The reason is simple: the μ-opioid receptor isn’t particularly “fussy” about what it binds. In pharmacology, the term promiscuous—minus the moral overtones—applies. The receptor can interact with a vast (effectively unlimited) number of chemically distinct molecules, including drugs that may not yet have been imagined, let alone synthesized.
In other words, the potential chemical space for opioid drugs is enormous, thanks to the power of synthetic organic chemistry.
Which brings us back to the analogy with microbial drug resistance.
Microbes evolve resistance because drugs eliminate susceptible organisms and leave resistant ones behind. The drug itself creates the evolutionary pressure that drives the emergence of resistant strains.
When law enforcement makes molecule X difficult to produce, distribute, or import, the market adapts by finding molecule Y. That molecule might be a slightly modified analog, a different chemical scaffold, or a completely new synthetic drug.
From a chemist’s perspective, this process looks strikingly similar to biological evolution. Under pressure from regulation and enforcement, the illicit drug market does what evolving organisms do: it searches chemical space for molecules that survive the new constraints. Some fail, some persist, and occasionally a compound emerges that is even more potent, easier to produce, or harder to detect than its predecessors. In effect, the global drug market functions like a vast chemical Petri dish, continuously generating new variants until one thrives under the latest conditions.
This is the iron law of prohibition: the harder the enforcement, the harder the drug. Here it is operating at the molecular level.
It is worth asking what we have gained from the massive effort required to tame fentanyl. Perhaps we have bought a temporary respite by suppressing one particularly dangerous drug.
But prohibition does not end the arms race—it accelerates it.
The process will almost certainly continue—just as predictably as microbial evolution in a Petri dish.



